- The paper presents a comparative analysis revealing that both methods capture state lifetimes of 100–150 ms.
- It demonstrates that HMMs identify more segmented temporal dynamics with less overlap than traditional microstates.
- The findings underscore the need for refined modeling techniques to better understand resting state EEG brain networks.
Resting State Brain Networks from EEG: Hidden Markov States vs. Classical Microstates
Introduction
The study explores the dynamics of resting state brain networks (RSNs), emphasizing two approaches using electroencephalography (EEG): Hidden Markov Models (HMMs) vs. classical EEG microstate analysis. Both methods demonstrate state lifetimes of 100-150 ms, suggesting similar temporal dynamics but revealing distinct spatial and temporal properties. The RSNs identified from the EEG are argued to be pivotal in understanding cognitive processes, with their millennium-long tradition as both a functional and clinical marker in neuroscience. The primary challenge lies in capturing rapid fluctuations, a task for which EEG's temporal resolution is particularly suitable compared to fMRI, despite the latter's superior spatial resolution.
Methodology
EEG Data Acquisition and Microstate Analysis
The study involved ten minutes of resting state EEG data recorded from six healthy subjects. The data was meticulously processed to remove artifacts before decomposition into principal components. Classical microstate analysis relies heavily on Global Field Power (GFP) time courses, where peaks are identified as local maxima, benefiting from high signal-to-noise ratios. Traditional clustering methodologies like k-means are employed to discern quasi-stable microstates, typically confined to four major clusters.
Hidden Markov Model Application
In contrast to microstates, the HMM offers a dynamic probabilistic model presenting the brain as transitioning between different states with Gaussian observations. This approach captures temporal and spatial dynamics potentially reflective of underlying physiological activities. HMMs applied to EEG data focus on state transitions using variational Bayes inference, allowing a tractable approximation of the posterior distribution. A consistent free energy decrease indicates patterns in data supporting a larger number of states, though focusing on four to ensure comparability with microstate analysis.
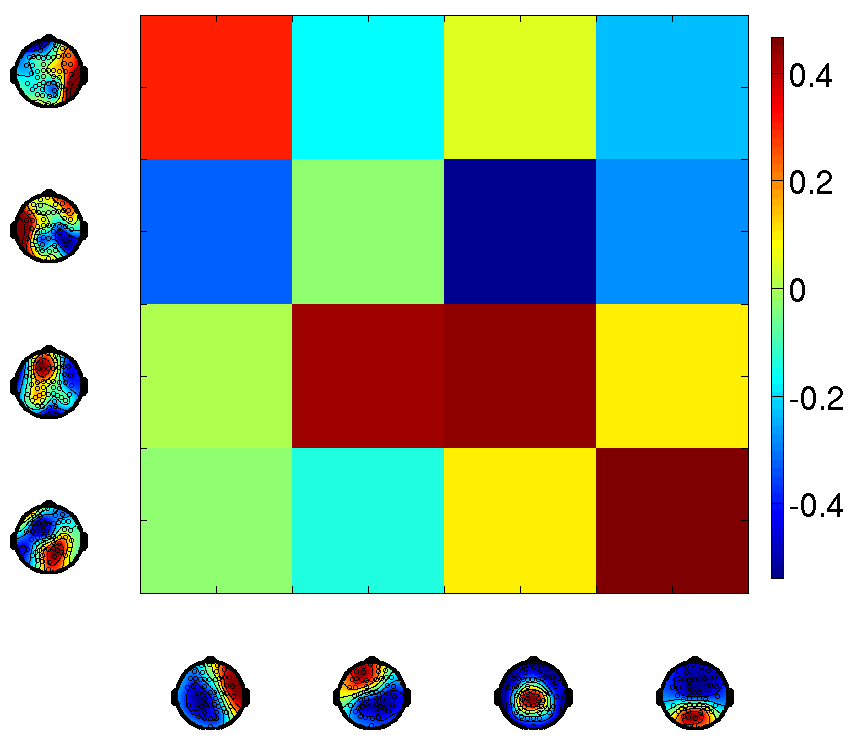
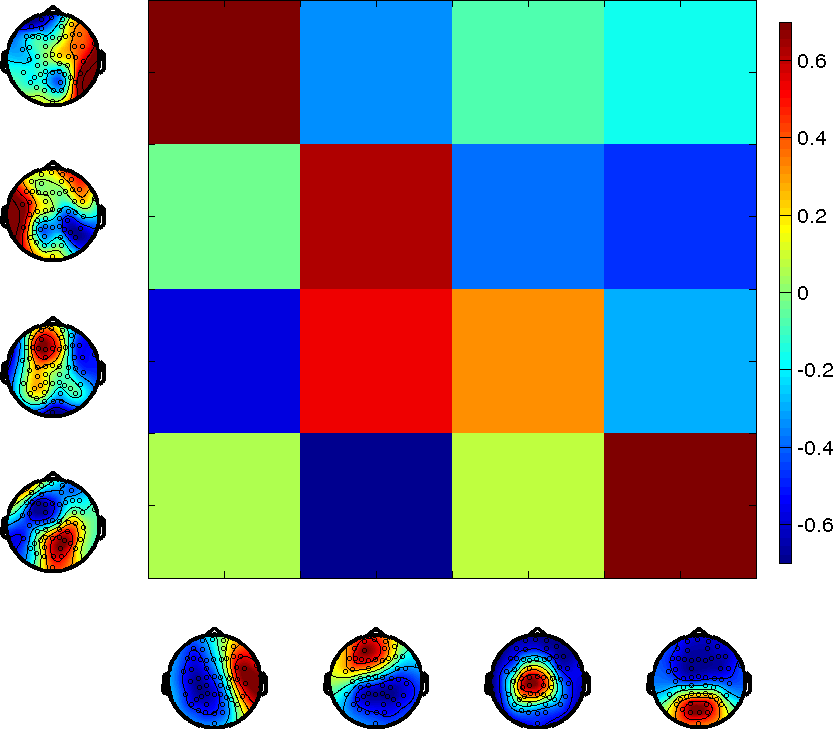
Figure 1: A detailed comparison of the spatial and temporal correlation structure between microstates and HMM states.
Comparative Analysis
Spatial and Temporal Properties
Classical microstates and HMM states share spatial activation regions but differ significantly in the temporal evolution of their time courses. Although pairwise correlations highlight similarities, the HMM format suggests states with less overlap and more significant negative correlations, opposing the homogeneous correlation structure of traditional microstates.
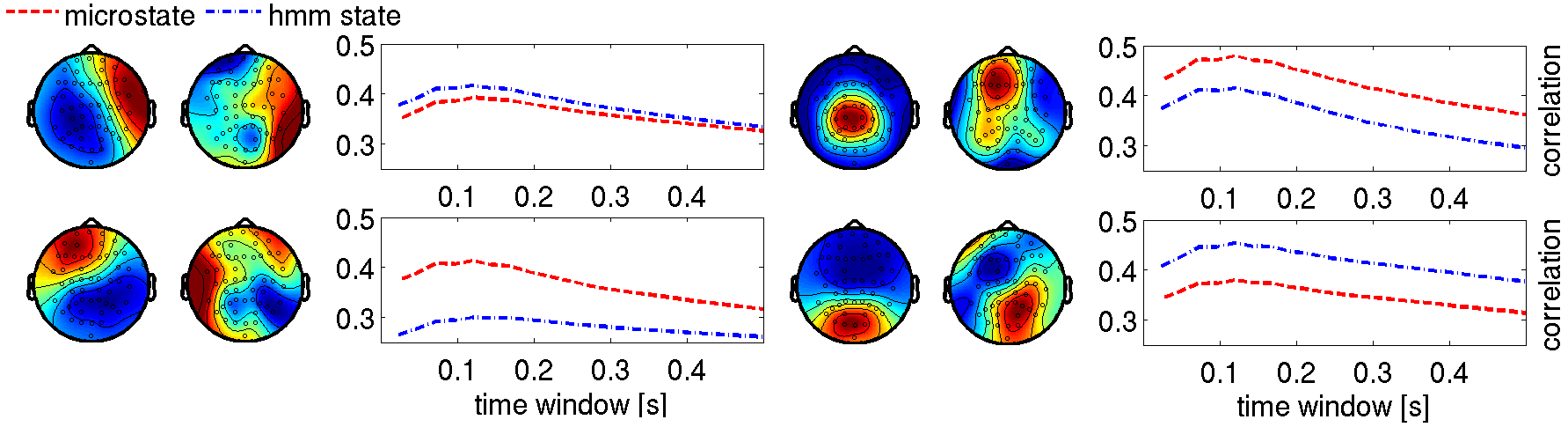
Figure 2: Microstates show strong positive temporal correlations across time courses, unlike the less correlated HMM states.
Scale and Correlation Analysis
Investigations into the temporal scale of state dynamics reveal dominant lifetimes aligning with classical findings, yet indicating the superior adaptability of HMMs in capturing the true dynamics of RSNs. The time scale analysis consistently shows maxima in correlations around 100–150 ms, agreeing with earlier observations. However, the stronger correlations amongst microstates suggest potential redundancy which might undermine the physiological accuracy.
Discussion and Conclusion
EEG-based analysis of RSNs via HMMs introduces an innovative, physiologically motivated alternative to classical microstate models. The temporal correlations derived from HMM suggest a more fragmented view of brain dynamics, which, albeit initially counterintuitive, may offer a closer representation of distinct cognitive processes. The study highlights the necessity for comprehensive evaluations, ensuring robust inference models that mitigate subject-specific variations and optimize the number of states modeled.
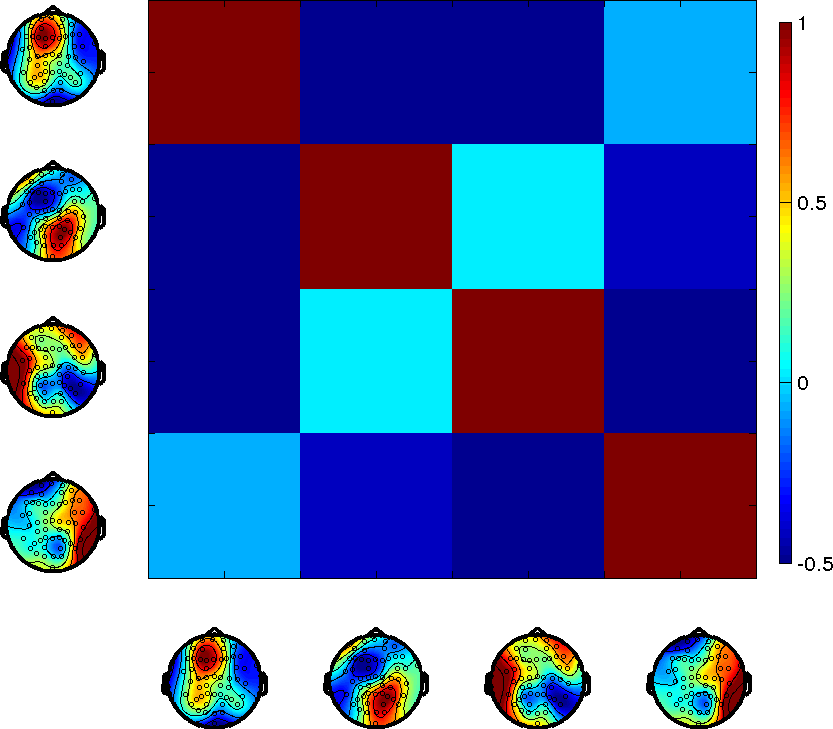
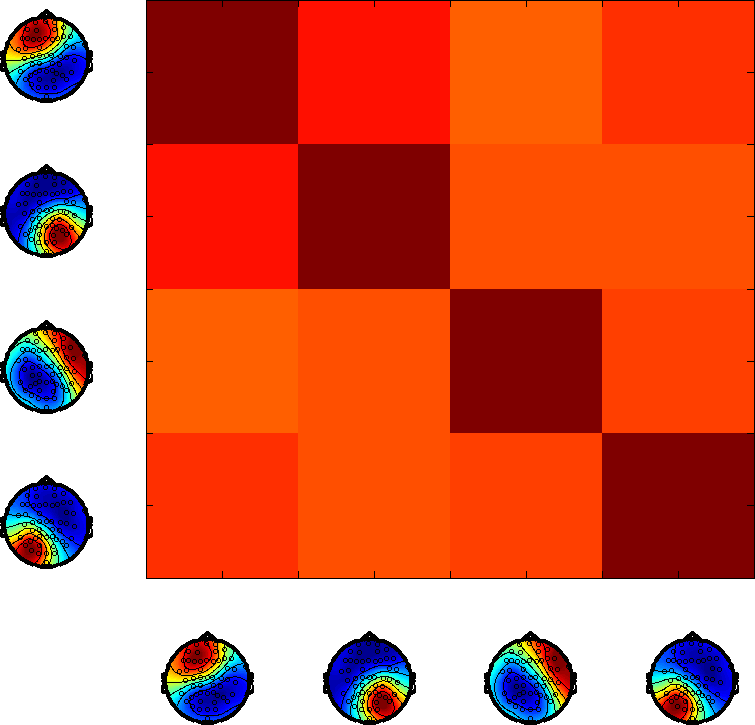
Figure 3: HMMs potentially capture long-range dependencies essential for effective cognitive processing, despite potential nonparametric limitations.
Further research is essential to refine HMM applications, addressing implicit criticisms on modeling core temporal dependencies, potentially culminating in dual-purpose models applicable across EEG and MEG. This model holds promise for greater insights into cognitive function, offering pathways for advancements in understanding brain physiology. The implications of this work underline the vital role robust modeling plays in bridging theoretical neuroscience with practical clinical applications. While HMM states potentially represent a greater accuracy in RSNs' physiology, collaboration across methodologies will be crucial in advancing both theory and application.