- The paper introduces a combined mechanistic and Gaussian Process framework to personalize hormonal cycle modeling.
- It employs a 13-dimensional delayed differential equation system to simulate hormone interactions across menstrual phases.
- GP models reliably predict menstrual cycle phases from noisy, downsampled data, highlighting their clinical applicability.
Towards Personalized Modeling of the Female Hormonal Cycle: Implementation and Insights
The paper "Towards Personalized Modeling of the Female Hormonal Cycle: Experiments with Mechanistic Models and Gaussian Processes" (1712.00117) presents a study focused on personalizing the modeling of the female hormonal cycle using a combination of mechanistic models and Gaussian Processes (GPs). This work is particularly relevant for healthcare applications, aiming to enhance predictions of menstrual cycle phases, identify hormonal disorders, and improve understanding of individual hormonal behaviors.
Mechanistic Modeling and Hormonal Data
The paper employs a mechanistic model, specifically a 13-dimensional delayed differential equation system, to simulate the hormonal cycle. This model captures the interactions of key reproductive hormones such as the luteinizing hormone (LH), follicle-stimulating hormone (FSH), estradiol (E2), progesterone (P4), and inhibin (Ih). It provides a high-fidelity representation of physiological dynamics across the follicular and luteal phases separated by ovulation. By varying model parameters—particularly those controlling LH synthesis—the researchers simulate diverse menstrual cycle patterns, accommodating individual variability.
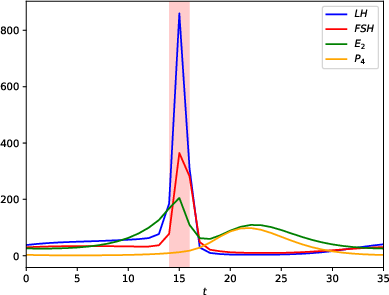
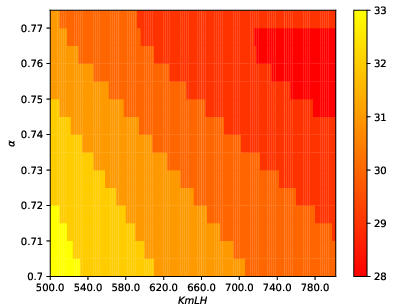
Figure 1: Evolution over time of the hormones of interest. Ovulation (in red) separates the follicular and luteal phases.
The in-silico data generated from the mechanistic model supports the Gaussian Process-based approach to predict hormonal cycle phases.
Gaussian Process Framework for Hormonal Prediction
The use of Gaussian Processes enables a non-parametric Bayesian framework tailored to capture the periodic and smooth nature of hormonal data. The kernel function, a combination of rational quadratic and periodic kernels, encapsulates these properties, optimizing predictions of hormonal levels over time.
Implementation Details
- Data Preprocessing: The raw output from the mechanistic model is perturbed with noise and downsampled to mimic realistic clinical scenarios. This involves adding Gaussian noise and varying sampling rates to the generated hormone levels.
- Training: Separate Gaussian Process Models (GPMs) are trained for each hormone, using time as the input variable. The models learn from both noise-augmented and downsampled data scenarios to estimate hormone levels and predict menstrual phases.
- Evaluation: Predictive performance is assessed by comparing GPM forecasts of hormonal peaks and troughs to ground truth data. Critical evaluation metrics include the ratio of correctly predicted days to actual cyclic phase lengths.
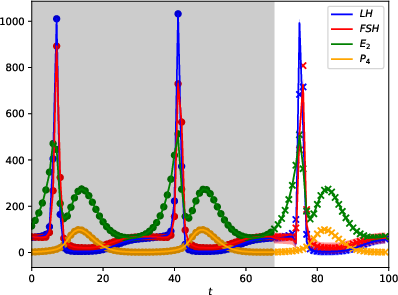
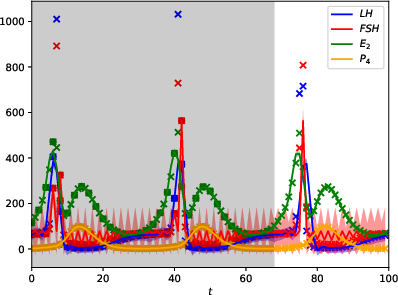
Figure 2: Daily training data.
The GPs demonstrate robust performance across varying data sampling frequencies and noise levels. Accurate predictions are contingent on the inclusion of hormonal peaks in the training set, highlighting the utility of strategic sampling. Data from Figures indicate that the GPMs can predict cycles accurately when trained with sufficient cyclic peaks, even under observation noise.
Practical Implications and Future Directions
The paper discusses the potential for these predictive models to improve diagnostics and tailor treatments for reproductive health issues. By reducing invasive hormone measurements through strategic data acquisition, GPMs present a viable path forward in hormonal cycle modeling.
Future work could extend to multi-output Gaussian Processes to capture interdependencies among hormones, as well as integrate phenotypic data from menstrual trackers. Such advancements would offer comprehensive models connecting hormonal dynamics with observable symptoms and behaviors.
Conclusion
Through the detailed examination of mechanistic models and Gaussian Processes, the paper demonstrates a robust approach for personalized modeling of the female hormonal cycle. This work sets the stage for improved healthcare applications by providing a foundation for precise hormonal predictions and enhanced understanding of menstrual cycle variability. Continued exploration in this domain promises to yield significant insights into women's health and hormonal disorder management.



