Non-invasive Blood Pressure Estimation Using Phonocardiogram
Abstract: This paper presents a novel approach based on pulse transit time (PTT) for the estimation of blood pressure (BP). In order to achieve this goal, a data acquisition hardware is designed for high-resolution sampling of phonocardiogram (PCG) and photoplethysmogram (PPG). These two signals can derive PTT values. Meanwhile, a force-sensing resistor (FSR) is placed under the cuff of the BP reference device to mark the moments of measurements accurately via recording instantaneous cuff pressure. For deriving the PTT-BP models, a calibration procedure including a supervised physical exercise is conducted for each individual. The proposed method is evaluated on 24 subjects. The final results prove that using PCG for PTT measurement alongside the proposed models, the BP can be estimated reliably. Since the use of PCG requires a minimal low-cost hardware, the proposed method enables ubiquitous BP estimation in portable healthcare devices.
Paper Prompts
Sign up for free to create and run prompts on this paper using GPT-5.
Top Community Prompts
Explain it Like I'm 14
What is this paper about?
This paper is about finding a simple, comfortable way to estimate a person’s blood pressure without using a squeezing arm cuff. The authors show that by listening to the heart with a tiny microphone (a phonocardiogram, or PCG) and measuring blood flow in the finger with a light sensor (a photoplethysmogram, or PPG), you can estimate blood pressure continuously and pretty accurately.
What questions did the researchers ask?
They wanted to know:
- Can we estimate blood pressure without a cuff by timing how fast a pulse travels through the body?
- Is using heart sounds (PCG) better than using electrical heart signals (ECG) for this timing?
- How accurate is this method for estimating the top number (systolic BP) and bottom number (diastolic BP)?
How did they do the study?
Signals they recorded
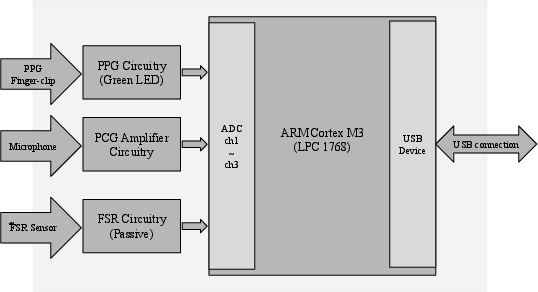
To make this work, they measured three things at the same time:
- Heart sounds (PCG): like placing a small microphone on the chest to hear the “lub-dub.”
- Finger blood flow (PPG): shining green light on the finger and seeing how much is reflected, which changes with each heartbeat.
- Cuff pressure (with a tiny pressure sensor called an FSR): to mark exactly when the standard BP monitor took each reading.
The key idea: Pulse Transit Time (PTT)
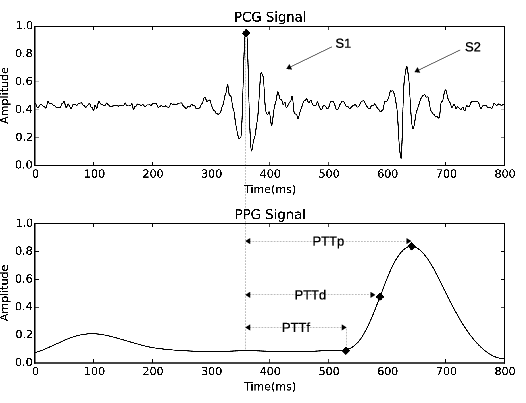
Think of your blood pulse like a wave that starts when your heart squeezes and then travels through your arteries to your finger. Pulse Transit Time (PTT) is the time it takes for that wave to go from the heart to the finger.
- When blood pressure is higher, arteries tend to be stiffer, and the pulse wave travels faster, so PTT is shorter.
- When blood pressure is lower, the wave travels slower, so PTT is longer.
Instead of using ECG (which needs sticky electrodes on the skin), the researchers used PCG, specifically the first heart sound (called S1), as the “start” time. This choice helps avoid extra delays that happen between the electrical signal of the heart and the actual mechanical action of pumping.
For the “arrival” time at the finger, they tried three points on the PPG signal:
- The start of the rise (called the “foot”)
- The steepest upward slope
- The peak of the pulse
Collecting and cleaning the data
They tested 24 healthy adults (ages 21–50). To make each person’s blood pressure change (so the model could learn), participants ran on a treadmill for about 3 minutes. Then they sat still while the researchers recorded PCG, PPG, and cuff readings.
They cleaned the signals to remove noise (like smoothing out a shaky video) and split the recordings into time chunks that matched each cuff measurement. They used the FSR pressure sensor under the cuff to pinpoint exactly when each official BP reading happened.
Building the model
Blood pressure and PTT are linked, but the exact link is different for each person. So they “calibrated” a simple model for every participant. In everyday terms, this means:
- Measure PTT and the real BP a few times (right after exercise, while BP is changing).
- Fit a simple equation that says: BP ≈ b0 + b1 × (1/PTT).
- Use most of the data to train the equation and a separate part to test it (so they don’t cheat by testing on the training data).
What did they find?
- Using the PPG peak or steepest slope worked best for timing; using the very start (“foot”) was less accurate.
- With the PPG peak as the arrival time, the average estimation errors were:
- About 7–8 mmHg for systolic BP (top number)
- About 3–4.5 mmHg for diastolic BP (bottom number)
- The estimates matched the real cuff readings quite well:
- Correlation about 0.84 for systolic and 0.86 for diastolic (closer to 1.0 is better)
In simple terms: their cuff-less method using a chest microphone and a finger light sensor tracked blood pressure changes reliably, especially for the bottom number.
Why does this matter?
- Comfort and convenience: No squeezing cuff means you can estimate BP more often and more comfortably.
- Continuous monitoring: You can watch how BP changes over time, which helps catch problems early, especially for people with high blood pressure (hypertension).
- Low cost and portability: A microphone for PCG is simple and cheap, and could even be built into phones or small wearables.
Final thoughts
This study shows a promising way to estimate blood pressure without a cuff by timing the pulse from the heart to the finger. Using heart sounds (PCG) as the start time is smart because it avoids delays that can make timing less accurate. The results suggest this approach could become part of everyday health monitoring devices. To make it ready for everyone, future work should test more people (including those with different health conditions), refine calibration to be easier or automatic, and ensure accuracy during daily activities.
Knowledge Gaps
Knowledge gaps, limitations, and open questions
Below is a concise list of what remains missing, uncertain, or unexplored in the paper, framed to guide future research:
- Generalizability: Results are based on 24 healthy adults (ages 21–50) post-exercise; performance in older adults, hypertensive patients, people with cardiovascular disease, and subjects with arrhythmias remains unknown.
- Calibration practicality: The subject-specific calibration relies on supervised exercise to perturb BP. It is unclear how practical this is outside lab settings, how often recalibration is needed, and how model parameters drift over hours, days, or across visits.
- Long-term stability: No assessment of temporal stability or repeatability of the PTT–BP model across different sessions, days, or physiological states (e.g., circadian variations, recovery, stress).
- Continuous monitoring: The pipeline evaluates PTT averaged over intervals anchored by cuff measurements (using FSR), but does not demonstrate beat-to-beat, real-time, cuff-less BP tracking between calibrations or during daily activities.
- Reliance on FSR during calibration: The method uses FSR under the cuff to identify reference measurement times; how the system operates and maintains accuracy without FSR in fully cuff-less, real-world use remains unaddressed.
- Model choice and justification: The paper adopts a linear inverse-PTT model (BP = b0 + b1·PTT⁻¹) despite earlier stating a nonlinear mechanistic relation; there is no systematic comparison of linear vs nonlinear fits per subject, nor analysis of when each is preferable (e.g., SBP vs DBP, different BP ranges).
- Physiological validity: The extent to which PCG S1 truly removes PEP is not quantified; validation against gold-standard markers of aortic valve opening (e.g., echocardiography, seismocardiography) is missing.
- PCG S1 detection robustness: The paper does not describe or evaluate algorithms to reliably distinguish S1 from S2 and detect S1 peaks under noise, motion, respiration, and different postures—critical for ambulatory use.
- PPG distal timing point selection: While PTTd and PTTp perform similarly, there is no analysis of robustness across conditions (e.g., vasoconstriction, temperature, perfusion changes, motion), nor guidance on when to prefer foot, max-slope, or peak.
- Path-length variability: The arterial path length (heart-to-finger) is not measured or modeled; how inter-individual differences (arm length, stature, vascular anatomy) impact PTT–BP mapping is not explored.
- Confounders affecting PPG: Effects of sympathetic tone, temperature, local vasomotion, and peripheral perfusion on PPG timing (and thus PTT) and the resulting BP error were not characterized.
- Side-specific effects: BP was measured on the right arm while PPG was recorded on the left finger; the impact of contralateral measurement and cuff inflation/deflation on systemic hemodynamics and PPG timing is not assessed.
- Error stratification: No analysis of estimation error as a function of BP range, heart rate, PTT range, or subject characteristics; subject-level variability and worst-case performance are not reported.
- Standards compliance: Accuracy is not benchmarked against clinical standards (e.g., AAMI/ISO 81060-2, BHS grading), leaving clinical feasibility and regulatory relevance unclear.
- Reference accuracy: The oscillometric device is taken as ground truth without reporting its validation in the dynamic post-exercise condition or quantifying its measurement uncertainty/bias.
- Motion and ambulatory scenarios: Data were collected with subjects seated, stable, and silent; robustness during common activities (walking, speaking, daily movement) is untested.
- Signal processing details: Key implementation details (filters beyond basic specs, S1/S2 classification, PPG fiducial detection, artifact rejection, quality metrics) are not provided, hindering reproducibility and targeted improvement.
- Wavelength and site dependence: Only green (530 nm) finger reflection PPG was used; the impact of PPG wavelength (IR vs red vs green) and alternative sites (ear, wrist, toe) on PTT–BP accuracy is not examined.
- Model inputs: The model ignores potentially informative covariates (e.g., heart rate, respiration, temperature, anthropometrics) that might reduce error or stabilize calibration.
- Posture and physiological states: Results are limited to seated, post-exercise recovery; performance across postures (supine/standing), breathing maneuvers, mental stress, and pharmacological influences is unknown.
- Hypertension range coverage: While SBP ranged up to 197 mmHg in post-exercise recovery, the dataset lacks controlled inclusion of sustained hypertensive states and hypotensive episodes; clinical extremes remain under-tested.
- Beat-level vs interval-level PTT: Averaging within intervals weighted near t3 may mask beat-to-beat variability; the sensitivity of results to window selection and weighting functions is not analyzed.
- PCG acquisition practicality: A chest-belt microphone was used; the feasibility, comfort, and signal quality of PCG acquisition with consumer devices (e.g., smartphone microphones, wearable stethoscope form factors) in everyday use are unaddressed.
- Head-to-head comparisons: No concurrent evaluation against ECG-based PAT/PTT on the same subjects to directly quantify the advantage of PCG over ECG in removing PEP and improving BP estimation.
- Dataset diversity and transparency: Limited demographic details (sex, BMI, ethnicity) and per-subject performance metrics are absent; code availability for processing and modeling is not stated.
- Regulatory and clinical pathways: The paper does not outline steps required to translate the method into clinically acceptable devices, including validation protocols, risk management, and user factors.
Practical Applications
Below are practical applications that flow from the paper’s findings and methods (PCG+PPG-based PTT for cuff-less BP, low-cost hardware, per-user calibration, and the signal-processing pipeline). Each item notes relevant sectors, potential tools/workflows, and key assumptions/dependencies.
Immediate Applications
- Cuff-less BP prototyping and benchmarking kits (academia, medtech R&D)
- What: Recreate the paper’s low-cost PCG (electret mic) + green PPG hardware and the PTTp/PTTd linear model as a reproducible research platform; use the shared dataset for benchmarking.
- Tools/workflows: 1 kHz sampling; PCG S1 detection; PPG peak/derivative timing; median + IIR band-pass filters; weighted averaging near measurement times; per-subject linear regression on PTT−1; Kaggle dataset (www.kaggle.com/mkachuee/noninvasivebp).
- Assumptions/dependencies: Quiet environment; stable chest microphone placement; individual calibration; healthy or near-healthy subjects; limited clinical generalizability.
- mHealth feasibility pilots for BP trend tracking (software, telehealth, wellness)
- What: Non-diagnostic trend monitoring between cuff readings using a phone camera PPG and a clip-on chest mic or chest-strap mic.
- Tools/workflows: Smartphone app with guided calibration session (short exercise to span BP), periodic cuff reference to reduce drift; alerting on deviations rather than absolute BP.
- Assumptions/dependencies: Motion/noise sensitivity; per-user calibration; regulatory labeling as “wellness/trend” (not for diagnosis).
- Fitness and recovery analytics using PTT trends (consumer health, sports tech)
- What: Chest-strap mic + finger PPG ring/watch to track training load, autonomic recovery, and stress via BP trend proxies.
- Tools/products: BLE-connected strap + wearable; in-app PTTp/PTTd models; session-based calibration (e.g., warm-up).
- Assumptions/dependencies: Non-clinical positioning; consistent sensor placement; users accept brief calibration; results interpreted as trends only.
- Augment remote monitoring with between-cuff BP estimates (telehealth)
- What: Provide continuous trend estimates between scheduled cuff measurements for home hypertension programs to flag changes for clinician review.
- Tools/workflows: Combine periodic cuff readings with ongoing PCG+PPG streams; weighted updates to subject-specific model; clinician dashboard showing trend bands and confidence intervals.
- Assumptions/dependencies: Periodic cuff measurements for recalibration; patient adherence; data privacy/security.
- Standardized calibration and labeling workflow for studies (academia, clinical research)
- What: Use the paper’s exercise-based calibration and FSR-under-cuff time-stamping to improve data quality in cuff-less BP studies.
- Tools/workflows: FSR to detect t1/t2/t3 cuff phases; mid-t3 windowing; weighted averaging near t3; supervised light exercise to induce BP variation.
- Assumptions/dependencies: Access to a cuff for reference; IRB and safety for exercise; population may be limited by exercise tolerance.
- Curriculum modules and teaching labs (education)
- What: Hands-on labs on cuff-less BP estimation, heart sound processing, and physiological modeling.
- Tools/workflows: Build the acquisition board; reproduce filtering and PTT feature choices (PTTd/PTTp outperform PTTf); evaluate MAE/STD/r.
- Assumptions/dependencies: Lab hardware budget; student-safe protocols; synthetic data for those without human subjects approval.
- Early-stage cohort explorations (academia, clinical pilot teams)
- What: IRB-approved pilot studies in narrowly defined cohorts (e.g., young adults, controlled hypertensives) to assess generalizability and failure modes.
- Tools/workflows: Protocolized posture control, sensor placement SOPs, repeated calibrations across days; Bland–Altman analyses.
- Assumptions/dependencies: Not yet suitable for clinical decision-making; cohort-specific optimization may be required.
Long-Term Applications
- Clinical-grade cuff-less BP wearables (healthcare devices)
- What: Chest patch (MEMS mic) + wrist or ear PPG for continuous, cuff-less BP with automatic calibration and drift management.
- Tools/products/workflows: Embedded PCG S1 detection; multi-point PPG timing; adaptive/transfer-learning calibration; on-device quality control; ISO 81060-3 compliant validation; integration with EHRs.
- Assumptions/dependencies: Robust motion/noise handling; population-wide calibration without exercise; regulatory clearance; performance across ages, skin tones, comorbidities, arrhythmias.
- Smartphone-only cuff-less BP for at-home management (software, digital therapeutics)
- What: Guided measurements using phone mic on chest and camera PPG, offering at-home, cuff-less spot checks and trend monitoring.
- Tools/workflows: Sensor alignment guidance, signal quality feedback, auto-calibration via occasional cuff; privacy-preserving on-device processing.
- Assumptions/dependencies: Acoustic coupling reliability; broad device heterogeneity; regulatory and clinical validation.
- Continuous in-car BP monitoring for safety and health (automotive, occupational health)
- What: Seatback acoustic sensors for PCG-like signals + steering wheel PPG to monitor BP trends during driving (fatigue/stress detection).
- Tools/workflows: Vibration/acoustics isolation; fusion with vehicle CAN data; edge inference; driver feedback/alerts.
- Assumptions/dependencies: Strong noise and motion artifacts; individualized calibration; liability and privacy considerations.
- Hospital and perioperative non-invasive BP trending (healthcare)
- What: Supplemental continuous BP trend monitoring on wards/ICU step-down as a bridge between cuff cycles, with alarms on rate-of-change.
- Tools/workflows: Integration with bedside monitors; auto QA (posture, placement, signal quality); clinician-configurable thresholds.
- Assumptions/dependencies: Interference from equipment; need for robust clinical validation and alarm safety standards.
- Maternal health and chronic disease programs (public health, policy)
- What: At-home cuff-less trend monitoring for preeclampsia risk and hypertension management, complementing less frequent cuff checks.
- Tools/workflows: Community health kits; cloud analytics for risk stratification; reimbursement pathways for remote patient monitoring.
- Assumptions/dependencies: Equity across body habitus and skin tones; usability training; clinical workflows for follow-up.
- Population-scale screening and epidemiology (policy, public health)
- What: Low-cost screening at scale (schools, workplaces, rural clinics) to detect BP trend outliers needing confirmatory cuff testing.
- Tools/workflows: Rapid setup stations using chest mic + finger PPG; centralized analytics; privacy-preserving data aggregation.
- Assumptions/dependencies: Clear triage protocols; avoidance of overdiagnosis; community buy-in; data governance.
- Calibration-free or minimal-calibration algorithms (software, AI/ML)
- What: Reduce or eliminate per-user exercise calibration using multi-sensor fusion (PCG+PPG+SCG/BCG), personalization layers, and transfer learning.
- Tools/workflows: Large, diverse labeled datasets; domain adaptation; physiological priors; on-device learning.
- Assumptions/dependencies: Access to high-quality data; mitigation of confounders (PEP variability, vascular tone, temperature).
- Standards, validation frameworks, and reimbursement pathways (policy, standards bodies)
- What: Develop test protocols for cuff-less PCG+PPG BP devices (posture, activity states, demographic diversity), and establish clinical utility and coding.
- Tools/workflows: Multi-site trials; consensus metrics beyond MAE/STD/r (clinical endpoints, trend accuracy); integration with ISO/AAMI frameworks.
- Assumptions/dependencies: Industry–academia–regulator collaboration; clarity on intended use (trend vs absolute).
- SDKs and silicon IP for edge BP estimation (semiconductors, IoT)
- What: Low-power firmware/SDKs implementing PCG+PPG PTT extraction, quality gating, and BP inference for wearables/IoT health devices.
- Tools/workflows: Hardware accelerators for filtering and peak detection; model update APIs; power and memory profiling.
- Assumptions/dependencies: Standardized sensor interfaces; robust cross-device performance.
- Novel form factors capturing heart sounds (hearables, textiles)
- What: In-ear or textile-integrated acoustic sensors to capture S1/S2 reliably for PTT without chest straps.
- Tools/workflows: Sensor design for acoustic coupling; noise suppression; comfort/usability studies.
- Assumptions/dependencies: Feasibility of consistent S1 detection away from the chest; motion and ambient noise robustness.
Key cross-cutting assumptions/dependencies impacting feasibility:
- Per-user calibration is currently required; removing it will likely demand multi-sensor fusion and advanced personalization.
- Accuracy demonstrated in 24 healthy adults; performance in elderly, hypertensives, arrhythmias, and diverse body types/skin tones remains to be established.
- Sensor placement stability and environmental noise critically affect PCG S1 detection; consumer-grade usability must address this.
- PPG quality varies with motion, perfusion, temperature, and skin tone; green-light PPG at 530 nm was used in the study.
- Regulatory requirements for absolute BP accuracy (e.g., ISO 81060-3) are stringent; near-term uses should emphasize trends and adjunct monitoring.
- Data privacy, security, and clinical workflow integration will determine adoption in healthcare settings.
Glossary
- Arterial tree: The branching network of arteries through which blood flows from the heart to peripheral tissues. Example: "one point of the arterial tree (proximal point) to another one (distal point) within the same cardiac cycle."
- Auscultatory: A blood pressure measurement method based on listening to arterial sounds (Korotkoff sounds) with a stethoscope. Example: "Currently, auscultatory and oscillometry are the most common approaches for non-invasively BP measurement."
- Ballistocardiogram (BCG): A recording of the body’s mechanical recoil due to cardiac ejection, used as a timing reference in cardiovascular monitoring. Example: "ballistocardiogram (BCG)"
- Band-pass filter: A signal processing filter that passes frequencies within a certain range and attenuates frequencies outside that range. Example: "IIR, Band-Pass"
- Bland-Altman plots: A method to assess agreement between two measurement techniques by plotting their differences against their averages. Example: "the Bland-Altman plots for estimations."
- Correlation coefficient (r): A statistical measure indicating the strength and direction of a linear relationship between estimated and reference values. Example: "correlation coefficient (r) of the estimation."
- Cuff-less: Refers to blood pressure measurement or estimation without using an inflatable arm cuff. Example: "a cuff-less non-invasive method, which can monitor BP continuously, would be desirable."
- Diastolic blood pressure (DBP): The minimum arterial pressure during relaxation and dilatation of the ventricles of the heart. Example: "its lower limit is called diastolic blood pressure (DBP)."
- Distal timing reference: The timing point at a downstream location (e.g., on PPG) used to compute transit time relative to an upstream (proximal) point. Example: "For the distal timing reference, a characteristic point on PPG, in the same cardiac cycle as the selected proximal point, is considered."
- Electrocardiogram (ECG): A recording of the heart’s electrical activity, commonly used as a proximal timing reference. Example: "exploit the electrocardiogram (ECG) and the photoplethysmogram (PPG) for the PTT calculation"
- Electret condenser microphone: A type of microphone using a permanently charged material, suitable for capturing heart sounds (PCG). Example: "a electret condenser microphone"
- Force-sensing resistor (FSR): A sensor whose resistance changes with applied force, used here to capture cuff pressure timing. Example: "a force-sensing resistor (FSR) is placed under the cuff of the BP reference device to mark the moments of measurements accurately via recording instantaneous cuff pressure."
- Infinite Impulse Response (IIR) filter: A digital filter with feedback, providing efficient frequency-selective filtering. Example: "IIR, Low-Pass"
- Leave-one-out (LOO): A cross-validation approach where each subject (or sample) is held out in turn for testing while the remainder are used for training. Example: "leave-one-out separation of the dataset to train and test sets is employed."
- Limits of agreement: The Bland-Altman boundaries within which most differences between two measurement methods lie. Example: "the limits of agreement (mean 1.96STD) are indicated"
- Low-pass filter: A filter that passes low-frequency components and attenuates high-frequency components. Example: "IIR, Low-Pass"
- Mean absolute error (MAE): The average of absolute differences between estimated and reference values, measuring accuracy. Example: "mean absolute error (MAE)"
- Myocardial contractility: The intrinsic ability of cardiac muscle to contract, influencing pre-ejection period and potentially PAT. Example: "myocardial contractility"
- Oscillometry: A cuff-based BP measurement method relying on pressure oscillations during cuff deflation. Example: "auscultatory and oscillometry are the most common approaches"
- Phonocardiogram (PCG): An acoustic recording of heart sounds, used here as a proximal timing reference for PTT. Example: "phonocardiogram (PCG)"
- Photoplethysmogram (PPG): An optical signal reflecting blood volume changes in tissue, used as a distal timing reference. Example: "photoplethysmogram (PPG)"
- Pre-ejection period (PEP): The interval between electrical activation and the opening of the aortic valve; included in PAT but not in PTT. Example: "pre-ejection period (PEP)"
- Proximal timing reference: The upstream cardiac event timing (e.g., S1 in PCG) used to start PTT measurement. Example: "can be considered as the proximal timing reference for measuring PTT"
- Pulse arrival time (PAT): The time from the ECG R-peak to a distal pulse feature; includes PEP and PTT. Example: "which is called pulse arrival time (PAT)"
- Pulse transit time (PTT): The time a pulse wave takes to travel between two arterial sites within the same cardiac cycle. Example: "pulse transit time (PTT)"
- Pulse wave velocity (PWV): The speed at which the blood pressure pulse propagates through the circulatory system. Example: "pulse wave velocity (PWV)"
- Reflection PPG: A PPG configuration where light reflected from tissue is measured rather than transmitted through it. Example: "reflection PPG using 530 nm (green) wavelength was captured"
- S1-peak: The prominent peak corresponding to the first heart sound in PCG, used as a proximal timing marker. Example: "PCG S1-peak, instead of ECG R-peak, can be considered as the proximal timing reference for measuring PTT"
- Standard deviation (STD): A measure of variability indicating how spread out estimation errors are. Example: "standard deviation (STD)"
- Systolic blood pressure (SBP): The maximum arterial pressure during heart contraction. Example: "The upper limit of BP is called systolic blood pressure (SBP)"
Collections
Sign up for free to add this paper to one or more collections.