- The paper presents a fully automated cascaded framework combining SVM-based artifact detection, wavelet packet decomposition, and a meta-heuristically optimized non-local means filter to clean EEG signals.
- It achieves high sensitivity (98.31%) and specificity (97.91%), effectively suppressing EMG artifacts while preserving underlying neuronal information.
- The approach outperforms existing methods on simulated and real data, ensuring minimal signal distortion and suitability for online EEG and BCI applications.
Introduction
Artifact contamination—primarily EMG-related muscle activity—remains a major constraint in single-channel EEG analysis, detrimentally impacting both clinical and BCI system performance. Conventional multi-channel blind source separation (BSS) approaches, ICA, or CCA-based methods, often degrade significantly for single-channel EEG due to strong source mixture and absence of suitable reference channels. Meanwhile, pure decomposition or wavelet methods either lack automation or compromise underlying neuronal information due to hard thresholding. The present work introduces a cascaded, fully automatic denoising framework that leverages WPD for multi-resolution time-frequency decomposition and a non-thresholded, parameter-optimized NLM filter for artifact suppression. Parameter optimization is tackled using GWO, yielding a systematic, signal-adaptive pipeline.
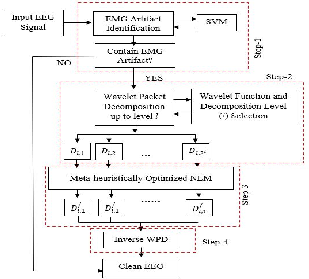
Figure 1: Basic architecture integrating SVM-based artifact detection, WPD decomposition, and NLM-based artifact correction with meta-heuristic optimization.
Methodology
Artifact Identification
A supervised SVM architecture discriminates corrupted (C-EEG) versus non-corrupted (NC-EEG) segments. Feature selection targets signal variance, Shannon entropy, and peak-to-peak amplitude, supporting robust classifier performance (sensitivity: 98.31%, specificity: 97.91%). Notably, the framework automates artifact recognition, bypassing the typical need for expert curation.

Figure 2: Experimental setup involving various cognitive and motoric tasks to induce EMG artifacts in EEG.
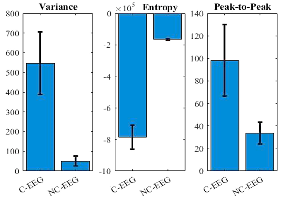
Figure 3: Discriminability of extracted features for SVM-based corrupted/clean EEG classification.
WPD-Based Multi-Resolution Decomposition
WPD is preferred over DWT and EMD-based methods for its consistent frequency band partitioning at each resolution, crucial for non-stationary EEG. Mother wavelet selection is formalized—minimizing reconstruction error identifies "fk6" as optimal. Shannon entropy criteria identify level 3 as the decomposition depth at which approximation coefficients cA contain less information than detail coefficients cD, ensuring effective scale selection.
Artifact coefficients are corrected via NLM, which computes patch-based weighted averages—capitalizing on non-local signal similarities. Unlike classical approaches, no arbitrary thresholding is present; instead, bandwidth (λ), patch width (P), and search width (M) are systematically explored. Empirical evaluation demonstrates that P=4 and M=50 optimize SAR and computation time.
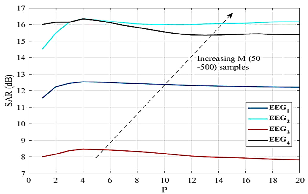
Figure 4: SAR improvement curves for varying patch widths and search sizes, highlighting the insensitivity of SAR to M but clear maxima over P=4.
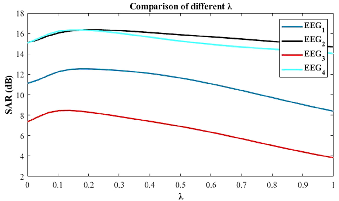
Figure 5: SAR sensitivity over λ values (bandwidth parameter), establishing the necessity of per-signal optimization.
Parameter λ is adaptively selected using the GWO meta-heuristic, empirically outperforming PSO optimizers in convergence and fitness.
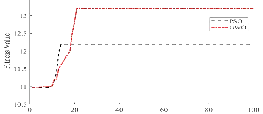
Figure 6: Superior convergence rate of GWO versus PSO for the λ search problem.
Signal Reconstruction
Inverse WPD synthesizes the artifact-corrected EEG from NLM-corrected coefficients, restoring the cleaned time series.
Experimental Results
Simulated Data
On simulated C-EEG (clean + EMG-contaminated), the pipeline accurately preserves both temporal and spectral structure (see below), greatly mitigating distortions observed in peer methods.
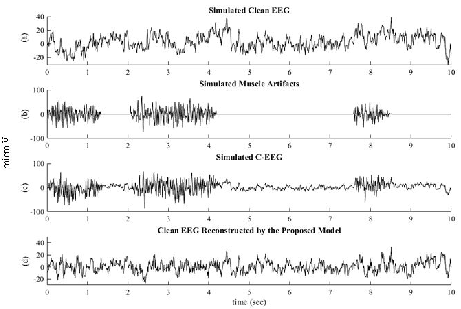
Figure 7: Simulated clean EEG, EMG, corrupted C-EEG, and corrected signal—demonstrating effective artifact removal and preservation of original EEG structure.
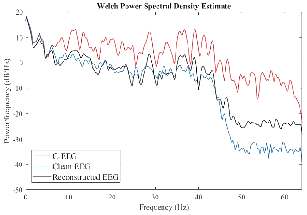
Figure 8: Power spectral densities of raw, contaminated, and reconstructed signals confirm restoration of canonical EEG bands post-denoising.
Comparison against MKNLMS-CS, tuneable WPD, and EEMD-CCA verifies the approach's ability to minimize information loss in clean intervals (mean CC = 0.8863 vs. 0.5089-0.5557 for other approaches) and achieve highest CC/SSIM over full signal (CC = 0.8675, SSIM = 0.6809).
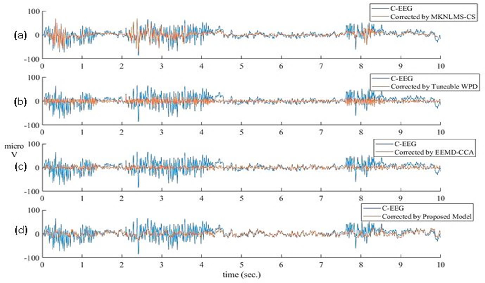
Figure 9: Visual comparison of denoised signals from referenced methods versus the proposed approach, highlighting minimal clean-region distortion.
Multi-Channel and Real Data Evaluation
Full 32-channel evaluation confirms scalability and robust generalization. Across all channels, mutual information (MI) retention is highest for the proposed method (average MI: 2.97 ± 0.70), consistently outperforming baseline denoising algorithms. The lack of manual tuning and persistence of information-theoretic metrics reinforce the suitability for online and unsupervised BCI contexts.
Theoretical and Practical Implications
The presented pipeline advances EEG artifact suppression in several dimensions:
- Automation: Eliminates ad hoc threshold selection and user intervention, achieving full pipeline automation.
- Generalization to Online/Real-Time Analysis: The parameter-free, meta-heuristic tuning and computational parsimony render the method practical for embedded and clinical EEG applications.
- Minimized Signal Distortion: Avoids over-smoothing and maintains high fidelity in clean epochs, supporting downstream pattern recognition or clinical diagnostics.
- Meta-Heuristic Optimization in Biomedical Signal Processing: Demonstrates the impact of global search-based optimizers for adaptive, context-specific parameter tuning in denoising filters; future extensions might integrate other evolutionary or swarm-based strategies.
- Extensibility: The cascade architecture accommodates extensions such as additional artifact types (EOG, ECG) via tailored classifiers or hierarchical denoising.
Conclusion
This work proposes a comprehensive, scalable, and robust artifact removal framework for single-channel EEG leveraging WPD decomposition, NLM-based denoising, and GWO-driven parameter optimization. Empirical analyses show clear quantitative and qualitative superiority over canonically used single-channel artifact removal baselines. The method's automation and high preservation of relevant EEG characteristics significantly enhance its applicability to real-world and online biomedical signal processing settings. Ongoing research directions include extension to multi-artifact scenarios and further reduction of computational overhead for real-time deployment.
(2201.01462)








