- The paper demonstrates that an AI-enhanced ECG model accurately predicts new-onset diabetes with an AUC of 0.80, surpassing conventional risk tests.
- It utilizes a deep convolutional neural network and pseudo-population adjustments to integrate ECG and demographic data for effective diabetes screening.
- The findings imply that wearable ECG devices could enable scalable screening, reducing the burden of undiagnosed diabetes in community settings.
New-Onset Diabetes Assessment Using AI-Enhanced ECG
Introduction
The study "New-Onset Diabetes Assessment Using Artificial Intelligence-Enhanced Electrocardiography" explores the efficacy of an AI-enhanced ECG model in predicting the likelihood of new-onset diabetes. This is a significant development given the increasing prevalence of diabetes and the limitations of current diagnostic measures. AI-enhanced ECG systems are positioned as valuable tools in early diabetes detection, leveraging the simplicity of ECG data acquisition through both clinical settings and wearable devices.
Methods
Study Population: The study involved a substantial dataset, consisting of 198,857 ECG records from 160,788 patients for training and validation, and 34,106 ECG records from 30,593 patients for testing. Participants were selected from a New York-based academic medical center's outpatient population, focusing specifically on individuals without prior diabetes.
Model Development: The authors implemented a deep convolutional neural network that integrates ECG data and demographic information to estimate HbA1c levels, using this to screen for diabetes in a general outpatient population. The pseudo-population model allowed for re-weighting of the dataset to represent the wider outpatient community (Figure 1).
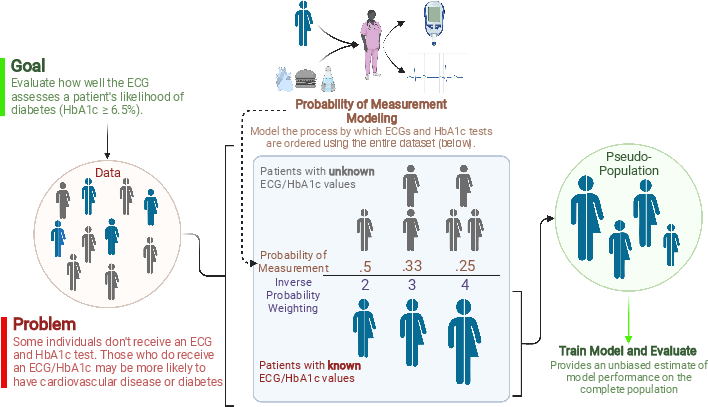
Figure 1: Diagram of pseudo-population construction.
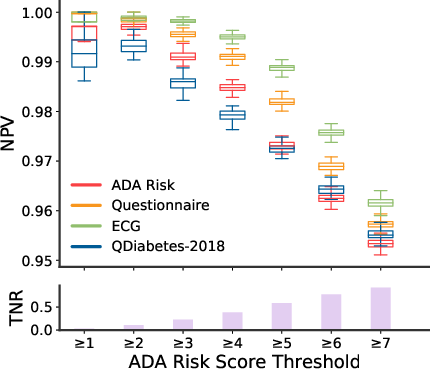
Evaluation Metrics: Key performance metrics included the area under the receiver operator curve (AUC) and positive predictive value (PPV). The comparisons were made against existing baselines like the ADA Risk Test and QDiabetes-2018.
Results
The AI-enhanced ECG model demonstrated superior performance in detecting new-onset diabetes across several metrics:
- Receiver Operator Curve (Figure 2): The model achieved an AUC of 0.80, significantly outperforming the ADA Risk Test and QDiabetes-2018, both of which had AUCs around 0.69.
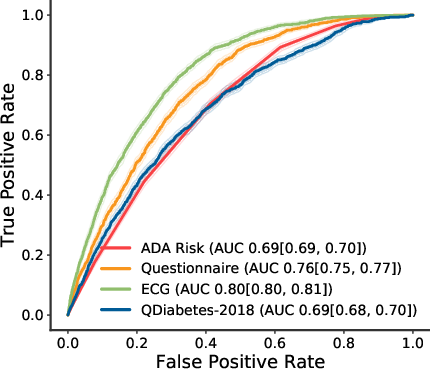
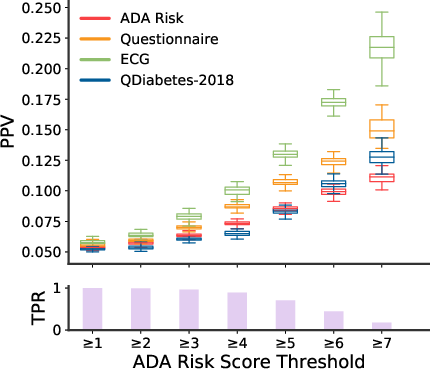
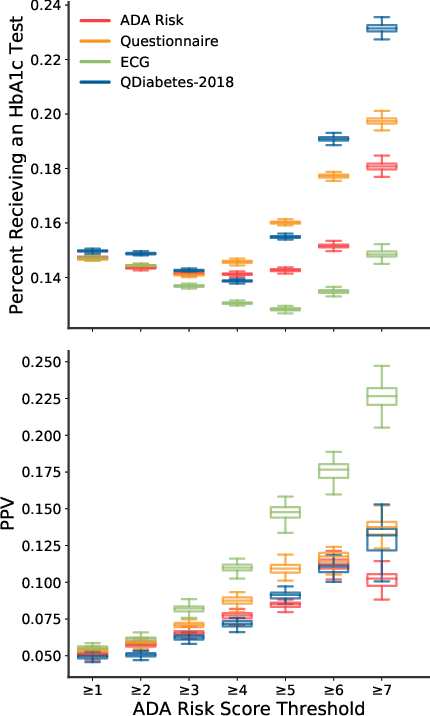
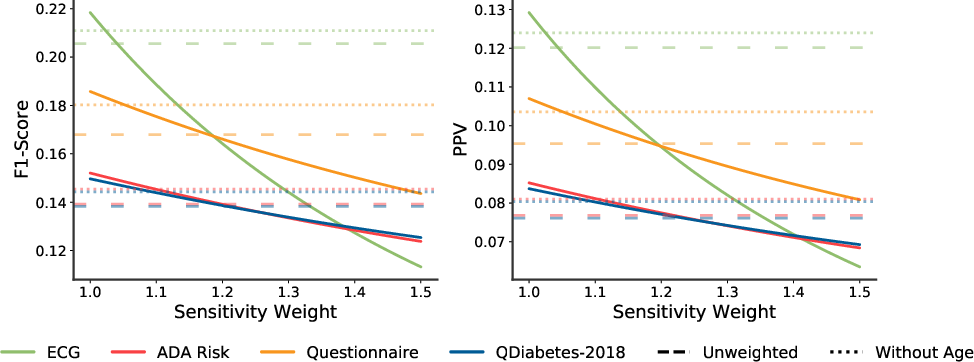
Figure 2: Receiver operator curve
Implications and Discussion
The study highlights the potential for AI-enhanced ECG models to improve screening for undiagnosed diabetes, addressing a critical gap in early disease detection. The ECG model can operate efficiently with single-lead ECGs, which are compatible with mobile wearable technology, expanding its applicability to community settings beyond traditional healthcare facilities. This capability aligns with the increasing demand for scalable diabetes screening solutions.
Despite robust performance metrics, the study acknowledges limitations related to data representation and model generalizability. The methodology involving pseudo-population construction ensures broader applicability yet requires further validation in prospective cohorts. Another notable point is the model's performance relative to human electrophysiologists, where it surpassed expert cardiologist interpretation, confirming the model's efficiency in subtle pattern recognition beyond current human capabilities.
Conclusion
The integration of AI with ECG data offers a promising pathway for widespread diabetes screening, potentially reducing the burden of undiagnosed diabetes on healthcare systems. Future research will explore further optimization of model architectures and expand its validation across diverse populations to ensure global applicability. This research represents a significant advancement in harnessing AI for better healthcare outcomes in chronic disease management.