Computational model advance deep brain stimulation for Parkinson's disease
Abstract: Deep brain stimulation(DBS)has become an effective intervention for advanced Parkinson's disease, but the exact mechanism of DBS is still unclear. In this review, we discuss the history of DBS, the anatomy and internal architecture of the basal ganglia(BG), the abnormal pathological changes of the BG in Parkinson's disease, and how computational models can help understand and advance DBS. We also describe two types of models:mathematical theoretical models and clinical predictive models. Mathematical theoretical models simulate neurons or neural networks of BG to shed light on the mechanistic principle underlying DBS, while clinical predictive models focus more on patients' outcomes, helping to adapt treatment plans for each patient and advance novel electrode designs. Finally, we provide insights and an outlook on future technologies.
Paper Prompts
Sign up for free to create and run prompts on this paper using GPT-5.
Top Community Prompts
Explain it Like I'm 14
A simple guide to “Computational models advance deep brain stimulation for Parkinson’s disease”
What this paper is about
This review explains how computer models help doctors and scientists improve deep brain stimulation (DBS), a treatment for Parkinson’s disease (PD). DBS is like a tiny brain pacemaker: an implanted electrode delivers gentle electrical pulses to help fix abnormal brain activity that causes movement problems. The paper shows how modeling the brain with computers can:
- Explore how and why DBS works
- Test safer, smarter ways to stimulate the brain
- Personalize treatment for each patient
- Inspire better electrode designs
What questions the paper asks
In everyday terms, the paper looks at four big questions:
- What goes wrong in the brain’s “movement control center” (the basal ganglia) in Parkinson’s?
- How might electrical stimulation change those faulty signals back toward normal?
- Which places in the brain, pulse shapes, and frequencies work best—and why?
- How can we use patient scans and math to plan surgery, adjust settings, and design better hardware?
How the researchers approach it (with simple analogies)
The review covers two kinds of computer models. Think of them like two different tools in a toolbox:
- Mathematical theoretical models:
- Neurons are modeled like tiny electrical circuits.
- Brain rhythms (like 13–30 Hz “beta waves”) are treated like musical beats that can go out of sync in PD.
- DBS is tested virtually to see how changing the “beat” affects movement signals.
- Clinical predictive models:
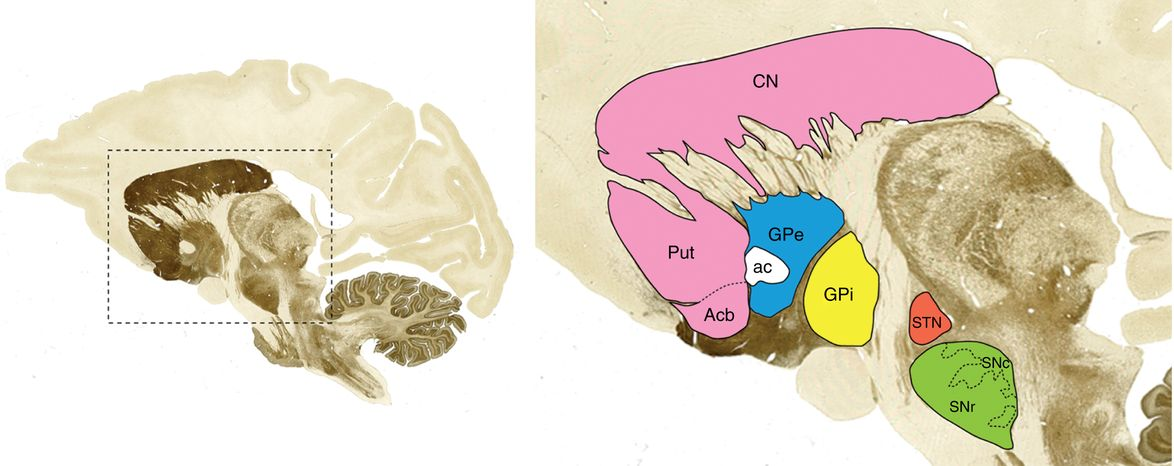
- Finite element models: a “digital sandbox” that simulates how electricity spreads through brain tissue.
- VTA (volume of tissue activated): the “bubble of influence” around the electrode showing what gets stimulated.
- Pathway Activation Models: a “road map” that predicts which nerve highways light up and how that links to symptoms and side effects.
- Software (e.g., Cicerone, LEAD-DBS) helps visualize electrodes in 3D, match them to brain anatomy, and predict outcomes.
The paper also reviews “closed-loop DBS,” which works like a thermostat:
- The system “listens” to brain signals (like beta rhythms) and automatically adjusts stimulation in real-time to keep activity in a healthy range.
What the models found and why it matters
Here are the main takeaways from the different modeling studies, and why they’re important:
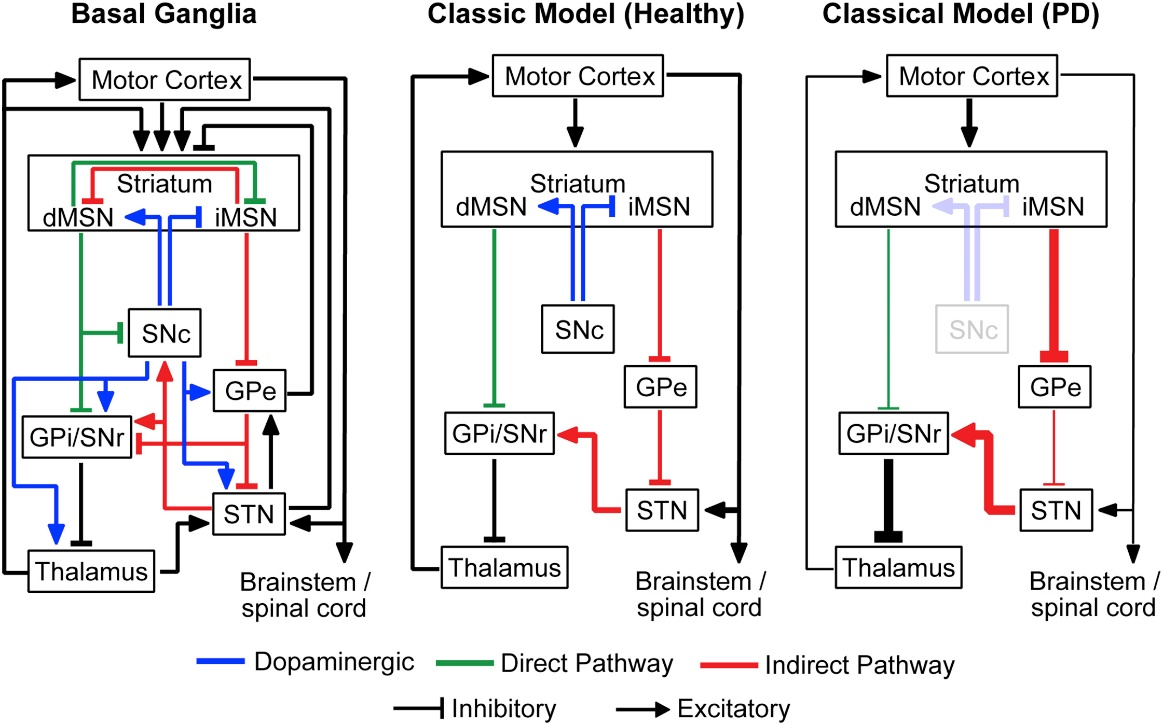
- DBS likely helps by “regularizing” bad rhythms, not just turning neurons up or down
- In PD, the basal ganglia can get stuck in unhealthy beta rhythms that slow movement.
- Models suggest DBS replaces these messy patterns with more regular signals—a bit like replacing static with a steady tone—so the brain’s “relay stations” can pass on movement commands more faithfully.
- This idea is called an “informational lesion”: rather than cutting tissue, DBS overwrites the noisy signal with cleaner timing.
- Frequency matters—a lot
- High-frequency stimulation (often above 100 Hz) tends to work best.
- Some models suggest this is because high frequencies match or disrupt the network’s “resonant frequency,” helping break the abnormal rhythm.
- Where you stimulate matters
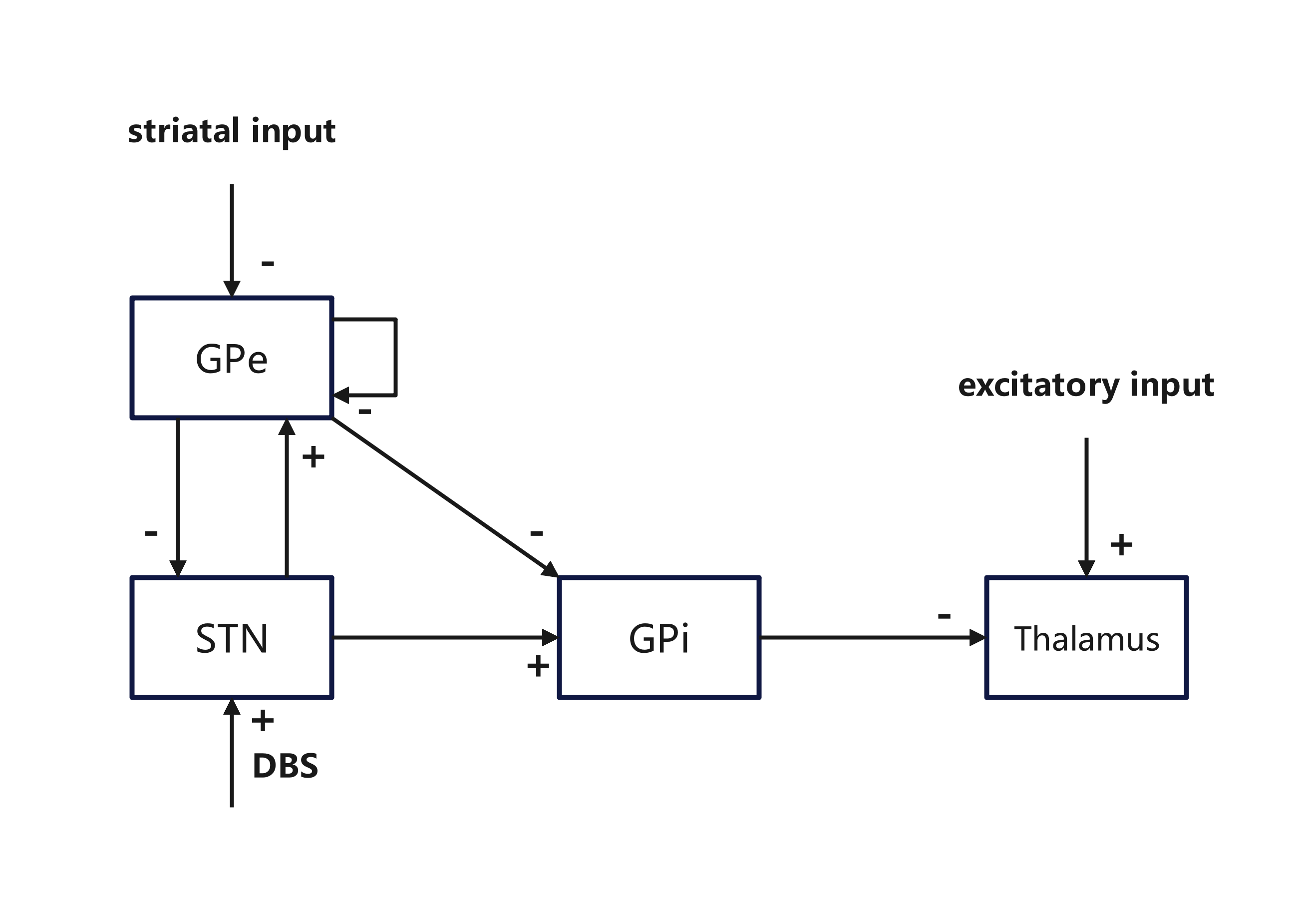
- The subthalamic nucleus (STN) is a common target. High-frequency STN stimulation often improves the “relay” of movement signals.
- Stimulating other areas (like GPe or GPi) can have different effects. Low-frequency stimulation can even make symptoms worse.
- Smarter pulse patterns can help
- Non-periodic patterns (not just steady ticking) and special designs like “coordinated reset” can desynchronize bad rhythms and sometimes keep benefits even after stopping stimulation.
- Genetic algorithms (computer search methods) can discover low-energy patterns that work as well as standard settings, which could save battery life and reduce side effects.
- Closed-loop (adaptive) DBS looks promising
- Using the brain’s own signals (like beta power) as a feedback marker lets the device adjust itself in real time.
- Models show closed-loop control can reduce symptoms with less energy than open-loop (always-on) stimulation.
- Patient-specific modeling can improve surgery and programming
- 3D tools built from MRI/CT help place electrodes more accurately and explain side effects.
- Modeling the VTA can guide parameter choices (how strong, which direction) to maximize benefit while avoiding nearby structures that cause side effects.
- Better electrode designs can “steer” current
- Directional (segmented) electrodes can aim stimulation like a flashlight beam instead of lighting up everything around.
- Models help compare designs, predict which shapes and contact layouts work best, and understand trade-offs (e.g., more detailed models take longer to compute).
Why this research matters for the future
- More precise understanding: Models turn complex brain activity into testable ideas, speeding up discoveries about how DBS works.
- Safer, faster care: Virtual “trial-and-error” can happen on a computer, reducing risk and time in the clinic.
- Personalized treatment: Patient-specific models + adaptive DBS can tailor therapy to each brain in real time.
- Better devices: Modeling guides smarter electrodes and energy-efficient pulse patterns, which may reduce side effects and extend battery life.
- Beyond Parkinson’s: The same ideas can be adapted for other brain disorders involving abnormal rhythms, like tremor or OCD.
In short, computational models are becoming powerful partners for doctors and engineers. They help explain what DBS does inside the brain, test new ideas before trying them in people, and move us toward more personalized, effective, and gentle treatments for Parkinson’s disease.
Knowledge Gaps
Knowledge gaps, limitations, and open questions
Below is a concise list of unresolved issues and uncertainties highlighted or implied by the review that future research could concretely address:
- Mechanism inconsistency: Do clinical benefits arise from regularizing GPi output (Rubin–Terman) or reducing GPi firing (Hahn–McIntyre)? Design experiments that simultaneously measure BG unit activity, LFPs, and behavior under controlled stimulation patterns to reconcile these conflicting mechanistic accounts.
- Frequency dependence: Several models fail to reproduce the clinically narrow effective frequency window (>100 Hz). Develop models that capture frequency-dependent entrainment, network resonance, and antidromic/collateral effects with realistic 3D electric fields.
- Origin of beta oscillations: The relative contributions of STN–GPe loops vs cortico–basal–thalamo circuits remain unresolved. Build multi-scale models that generate and suppress beta under dopamine depletion and validate against multi-site recordings.
- Soma versus axon activation: The “informational lesion” hypothesis (soma inhibition with axon activation) lacks direct human validation. Perform intraoperative paradigms to dissociate somatic and axonal contributions with paired stimulation–recording and pathway-specific markers.
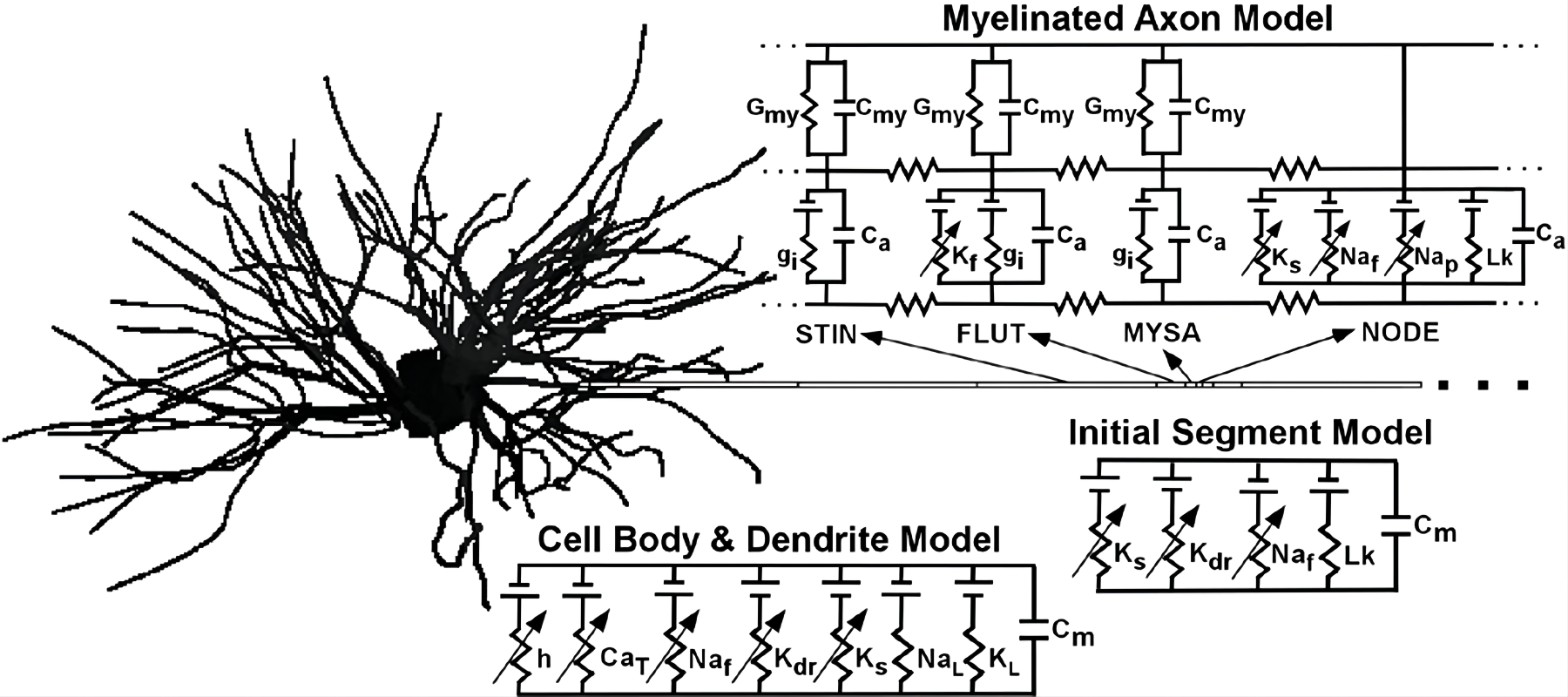
- Oversimplified BG heterogeneity: Many models assume homogeneous neuron populations and uniform STN activation. Incorporate neuron subtype diversity, dendritic morphology, short-term synaptic dynamics, and spatially heterogeneous microanatomy into 3D network models.
- Missing 3D biophysics: Early models ignore electrode orientation, anisotropic/inhomogeneous tissue conductivities, and complex field distributions. Standardize 3D finite-element pipelines with validated conductivity tensors and electrode geometries.
- Uncertain VTA ground truth: Volume of tissue activated estimates vary widely across volume-conductor (VC) models. Establish ground truth via intraoperative threshold mapping, microstimulation, and chronic sensing to calibrate VTA predictions.
- Pathway Activation Model (PAM) limits: PAM neglects axonal branching complexity, synaptic interactions, and network-level dynamics. Extend PAM to hybrid pathway–network models linking axonal recruitment to downstream network activity and behavior.
- Tractography uncertainty: DWI tractography has false positives/negatives that can misguide targeting. Quantify and propagate tractography uncertainty into outcome predictions and controller design.
- Biomarker adequacy for closed-loop DBS: Reliance on beta power alone is fragile across medication/sleep states and artifacts. Develop multi-biomarker frameworks (e.g., phase–amplitude coupling, spiking surrogates, kinematics) with adaptive state-dependent thresholds.
- Closed-loop control robustness: Comparative performance, stability guarantees, and energy costs of PI, fractional, MPC, and ML controllers under implant constraints (latency, compute, power) are not established. Benchmark controllers on standardized in silico “virtual patient” suites and in-human feasibility.
- PRC-based control translation: Phase response curve estimation and phasic burst protocols are mostly model-level. Demonstrate in-human PRC estimation, state dependence, and efficacy/safety of PRC-optimized stimuli.
- Non-periodic/patterned stimulation (e.g., CR): Long-term efficacy, generalization of parameters, and safety across patients remain uncertain. Run head-to-head trials versus standard DBS and test STDP-based mechanisms in human circuits with chronic sensing.
- Alternative targets (GPe, GPi, multi-target): Model predictions for efficacy at non-STN targets are not rigorously matched to clinical outcomes. Integrate collateral and afferent modulation in simulations and test model-derived parameters in controlled trials.
- Linking neural metrics to behavior: Surrogates like thalamic relay error and beta power are not standardized behavioral correlates. Define and validate model-to-behavior mappings (e.g., kinematic metrics) across tasks and clinical states.
- Non-motor domains: Current models underrepresent circuits mediating cognition, mood, speech, and autonomic function. Build network models predicting non-motor outcomes and side-effect thresholds to guide programming.
- Disease progression and variability: Few models capture longitudinal dopamine loss, compensatory plasticity, age, and comorbidities. Create longitudinal, patient-specific models that update with repeated sensing/imaging.
- Electrode–tissue interface dynamics: Time-varying impedance, gliosis, micromotion, and encapsulation are rarely modeled. Quantify their long-term effects on fields, activation thresholds, and closed-loop stability.
- Energy–efficacy trade-offs: Multi-objective optimization of symptom relief, side effects, and battery life for novel patterns (e.g., burst, non-periodic) is underexplored. Develop optimization frameworks with patient-specific constraints.
- Standardization and reproducibility: Lack of public datasets, benchmark tasks, and open code impedes comparison. Establish community challenges with shared MER/LFP/imaging/stimulation-response datasets and reference models.
- Identifiability and uncertainty: Many models are heavily tuned without sensitivity or uncertainty analyses. Apply Bayesian inference, global sensitivity analysis, and report parameter posteriors/credibility to assess robustness.
- Directional lead modeling: Divergent VC representations of segmented electrodes yield inconsistent activation predictions. Define consensus modeling practices validated against benchtop field mapping and in vivo thresholds/impedances.
- Sensing during stimulation: LFPs recorded under DBS are contaminated by artifacts. Develop validated artifact-suppression pipelines and hardware strategies to enable reliable closed-loop biomarkers.
- Ethical/regulatory path for adaptive DBS: Model-guided parameter search and autonomous control lack clear validation criteria. Specify safety/stability proofs, monitoring, and trial designs acceptable to regulators.
- Cross-scale integration: Few frameworks connect ion-channel kinetics, single-cell dynamics, mesoscale networks, and whole-brain connectomics to explain DBS effects end-to-end. Build modular multiscale co-simulation platforms with calibration at each scale.
- Cross-species translation: Discrepancies between rodent/primate and human findings (e.g., optogenetic STN stimulation) are unresolved. Create species-specific parameterizations and cross-species validation protocols.
- Surgical reality gaps: Planning tools often neglect brain shift, CSF loss, and post-op lead migration. Incorporate probabilistic lead reconstruction and intraoperative updates into models and outcome predictions.
- Predicting adverse effects: Quantitative models linking VTA/pathway activation to specific side effects (speech, mood, paresthesia, oculomotor) and their thresholds are limited. Build side-effect atlases with pathway-level activation models.
Collections
Sign up for free to add this paper to one or more collections.