- The paper introduces a CNF method to efficiently sample from the posterior distribution, enabling rapid, high-fidelity MR image reconstructions.
- The study demonstrates improved PSNR and SSIM over CGANs and Langevin samplers using the fastMRI dataset.
- The approach generates pixel-wise uncertainty maps, offering actionable diagnostic insights and potential for clinical application.
Comprehensive Analysis of "A Conditional Normalizing Flow for Accelerated Multi-Coil MR Imaging"
Introduction and Motivation
The field of magnetic resonance imaging (MRI) primarily seeks to optimize the trade-off between imaging quality and acquisition time. Traditional MRI techniques, while capable of producing high-resolution images, often suffer from extended acquisition durations leading to patient discomfort and increased probabilities of artifacts. In response, accelerated MRI methods, which attempt to reduce scan times by undersampling k-space data, present a challenging ill-posed inverse problem requiring sophisticated reconstruction techniques. The paper "A Conditional Normalizing Flow for Accelerated Multi-Coil MR Imaging" (2306.01630) addresses this by proposing a novel methodology for posterior sampling using Conditional Normalizing Flows (CNFs). This method offers a way to recover detailed images with a quantifiable measure of uncertainty, thus enhancing both clinical diagnostics and machine learning approaches in MRI.
Methodology
The study introduces a CNF tailored for accelerated MRI, capable of efficiently sampling from the posterior distribution of image reconstructions. This approach directly confronts the challenge posed by the ill-posed nature of inverse problems in undersampled MRI by utilizing the benefits of Bayesian techniques to provide multiple plausible reconstructions.
Measurement Model: The work models multi-coil MRI measurements in k-space, where each coil contributes information regarding the underlying anatomical structures. This model emphasizes the signal's transformation through Fourier space and highlights the constraints imposed by the acquisition process.
Conditional Normalizing Flow (CNF): The central innovation in the paper is the application of CNFs, which are generative models learned to map a simple latent distribution to the target data space using invertible transformations. The CNF designed in this research incorporates a conditioning network that processes zero-filled image estimates from undersampled data to guide the CNF in its mapping task (Figure 1).
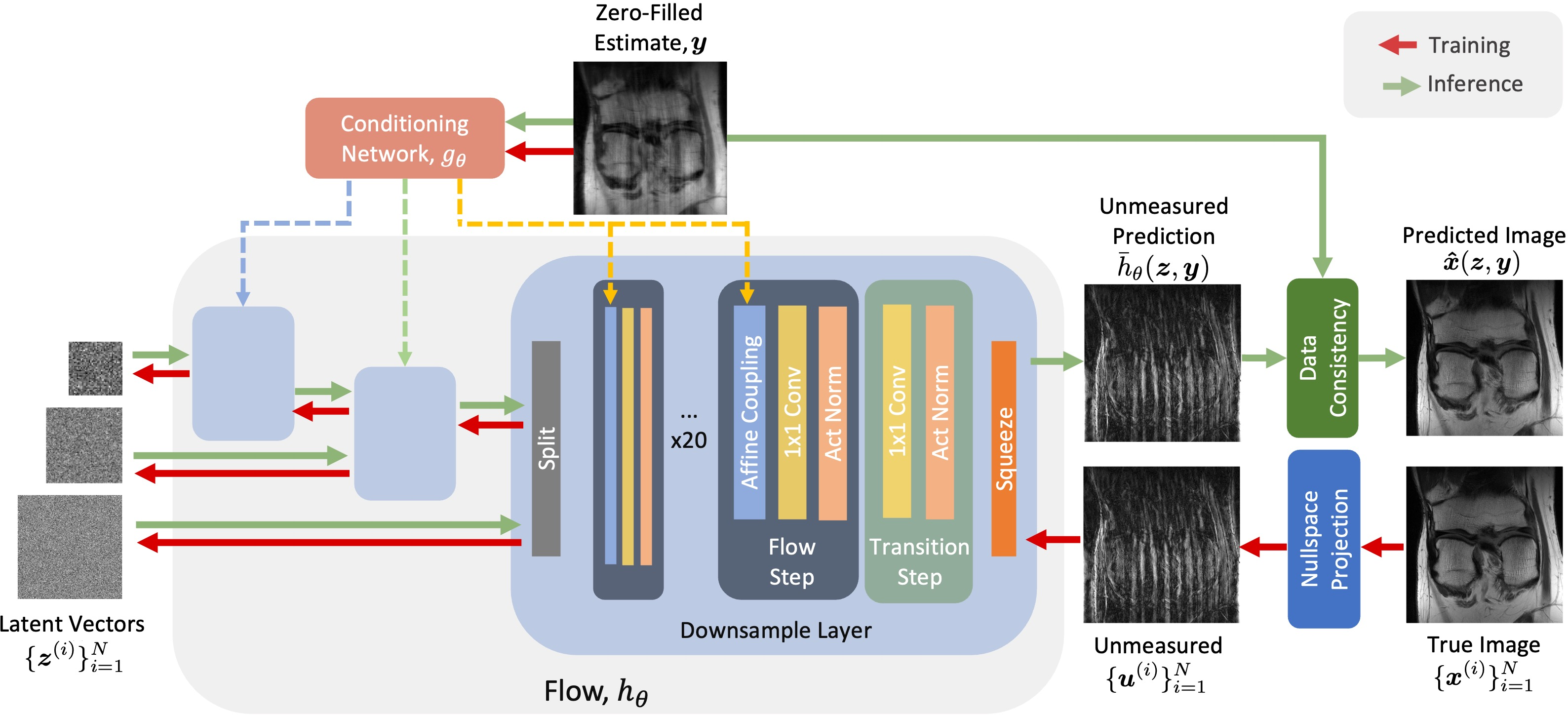
Figure 1: The architecture of our CNF. The conditioning network gθ takes in multi-coil zero-filled image estimates y and outputs features used by the flow model hθ. The flow learns an invertible mapping between Gaussian random samples zi and images ui that are the projections of the training images xi onto the non-measured subspace.
Results and Evaluation
Utilizing the fastMRI dataset, the proposed method demonstrates superior performance over existing state-of-the-art approaches like CGANs and Langevin-based samplers. Key metrics such as peak signal-to-noise ratio (PSNR) and structural similarity index (SSIM) were used to evaluate the performance.
Quantitative and Qualitative Analysis: The CNF showed noteworthy improvements in PSNR and SSIM, attaining faster inference times while maintaining high reconstruction fidelity (Tables 1 and 2). The method's ability to provide posterior samples facilitates the construction of pixel-wise uncertainty maps, offering intuitively visual tools for determining reconstruction reliability (Figures 2 and 3).
Visual Results: Figures illustrate the model's capacity to capture structural variations (Figure 2), highlighting the clinical and practical potential of CNFs in developing robust imaging solutions for dynamic and complex tasks in real-world usage scenarios.
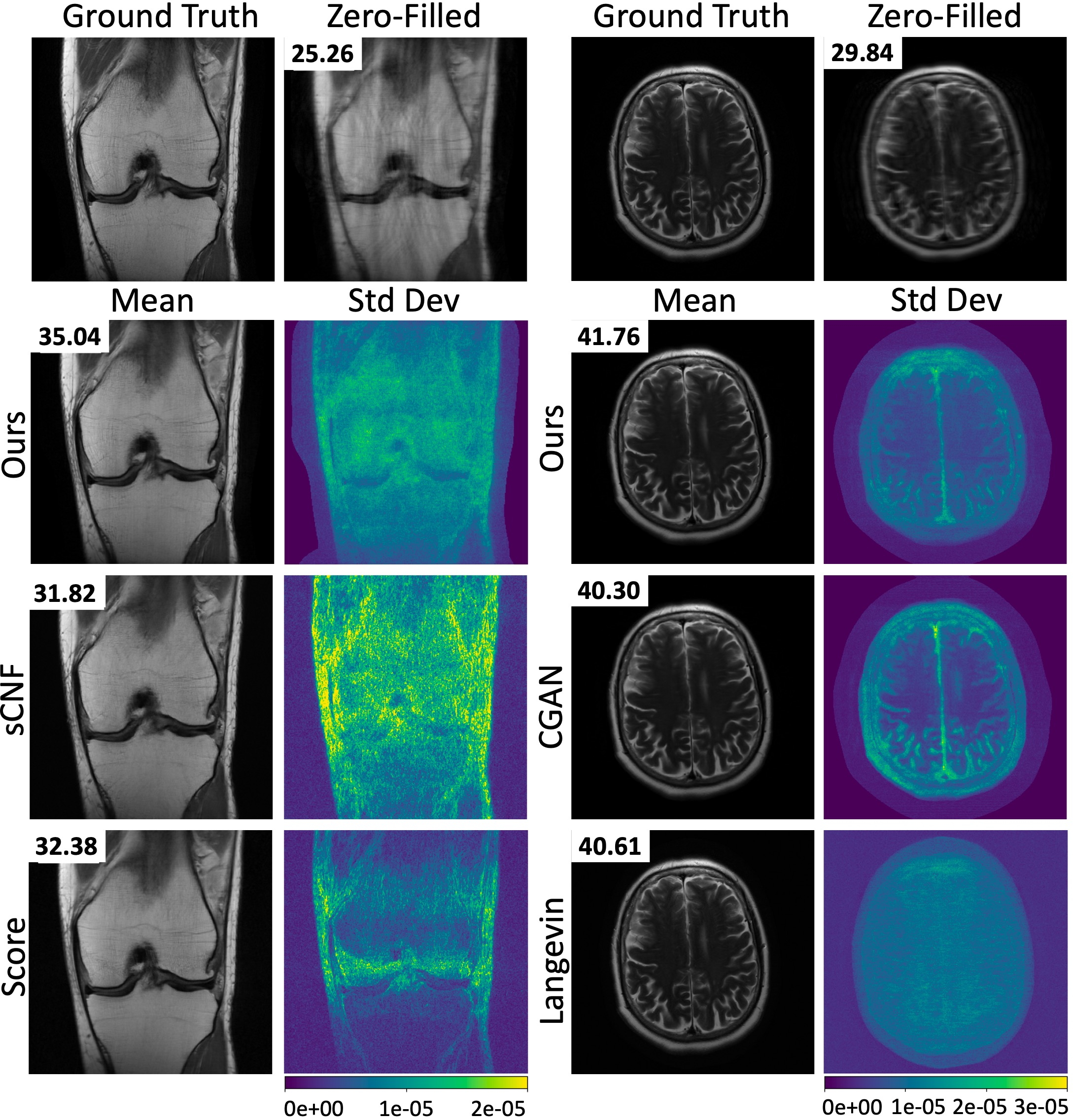
Figure 3: Mean images and pixel-wise standard-deviation maps computed from 8 and 32 posterior samples for the brain images and knee datasets, respectively. The standard-deviation maps show which pixels have the greatest reconstruction uncertainty. The corresponding PSNR is shown on each reconstruction.
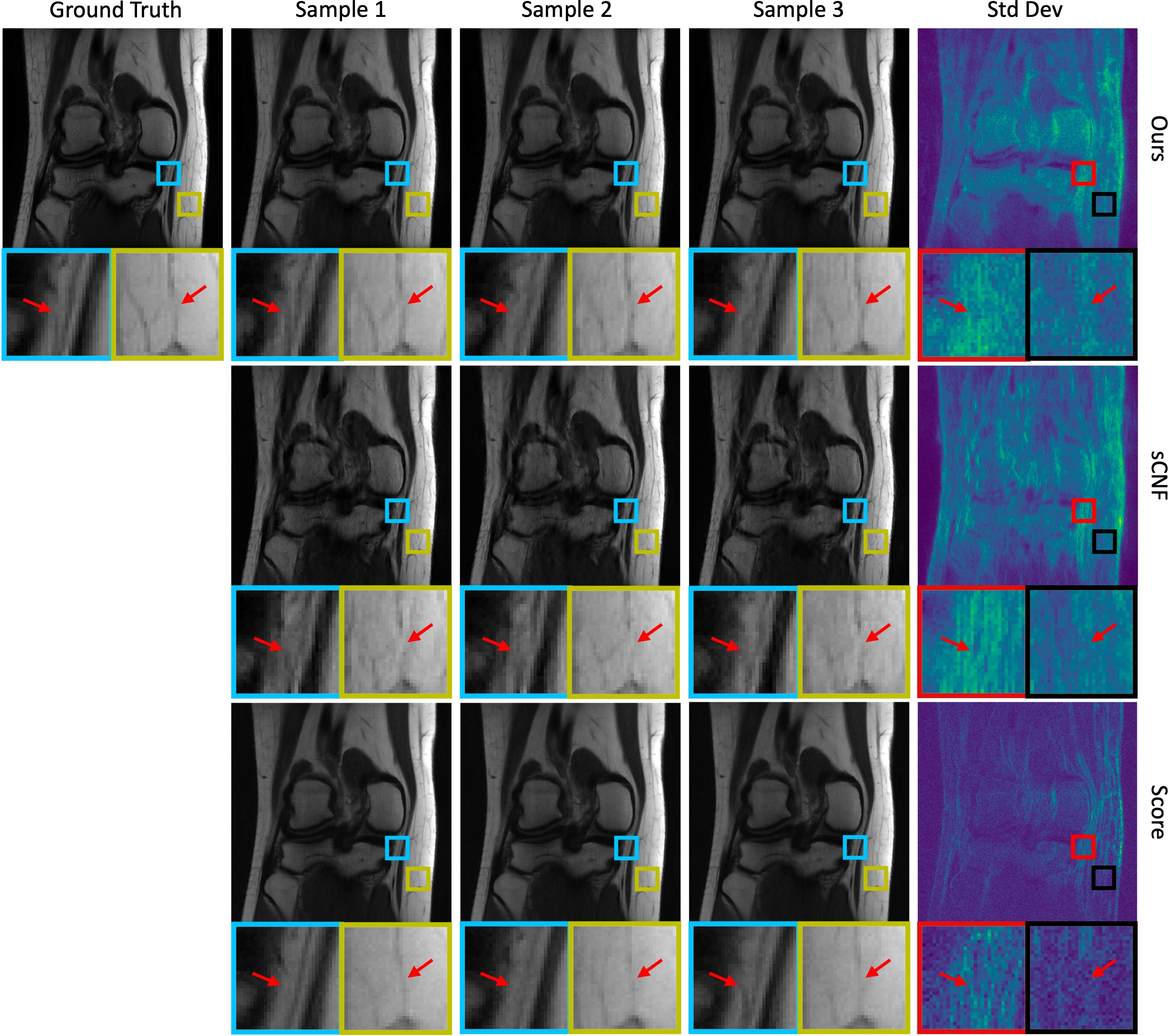
Figure 2: Examples of posterior samples and standard-deviation maps for the knee data. The samples show important structural variations. This demonstrates the advantages of generating multiple reconstructions and computing a pixel-wise standard-deviation map.
Implications and Future Research
The study's findings bring forward several critical implications for both theoretical and practical domains in MRI technology. Firstly, it illustrates the feasibility of CNFs for real-time clinical applications by showcasing rapid sampling and maintaining high reconstruction quality, paving the way for widespread adoption. Secondly, the framework's flexibility offers potential applicability to other inverse problems or enhanced extensions in machine learning models dealing with high-dimensional data.
Future research could explore the integration of this CNF approach with more advanced architectures or hybrid systems that amalgamate several generative models, fostering improvements in efficiency and performance. Additionally, exploring the direct applications of the uncertainty maps in clinical diagnostics could provide richer insights into practical utility, potentially influencing MRI protocols that prioritize patient safety and comfort without sacrificing diagnostic accuracy.
Conclusion
In summary, the development of a Conditional Normalizing Flow tailored for multi-coil accelerated MRI represents a significant advance in the field of medical imaging. By leveraging the strengths of Bayesian inference and normalizing flows, this approach not only achieves superior image reconstruction but also provides vital uncertainty quantification, thereby offering transformative insights for clinical and research applications in MRI.
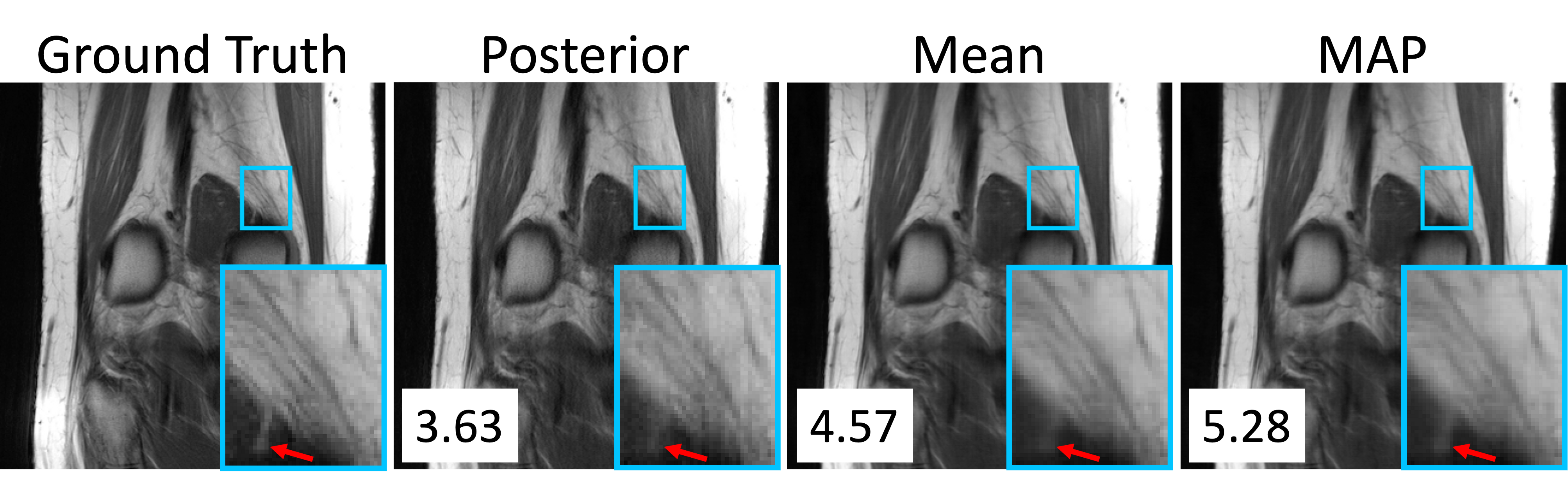
Figure 4: Examples of a ground-truth image, one posterior sample, an average of P=8 posterior samples, and a MAP estimate. The log posterior density in units of bits-per-dimension is shown in the bottom right corner of each image.



