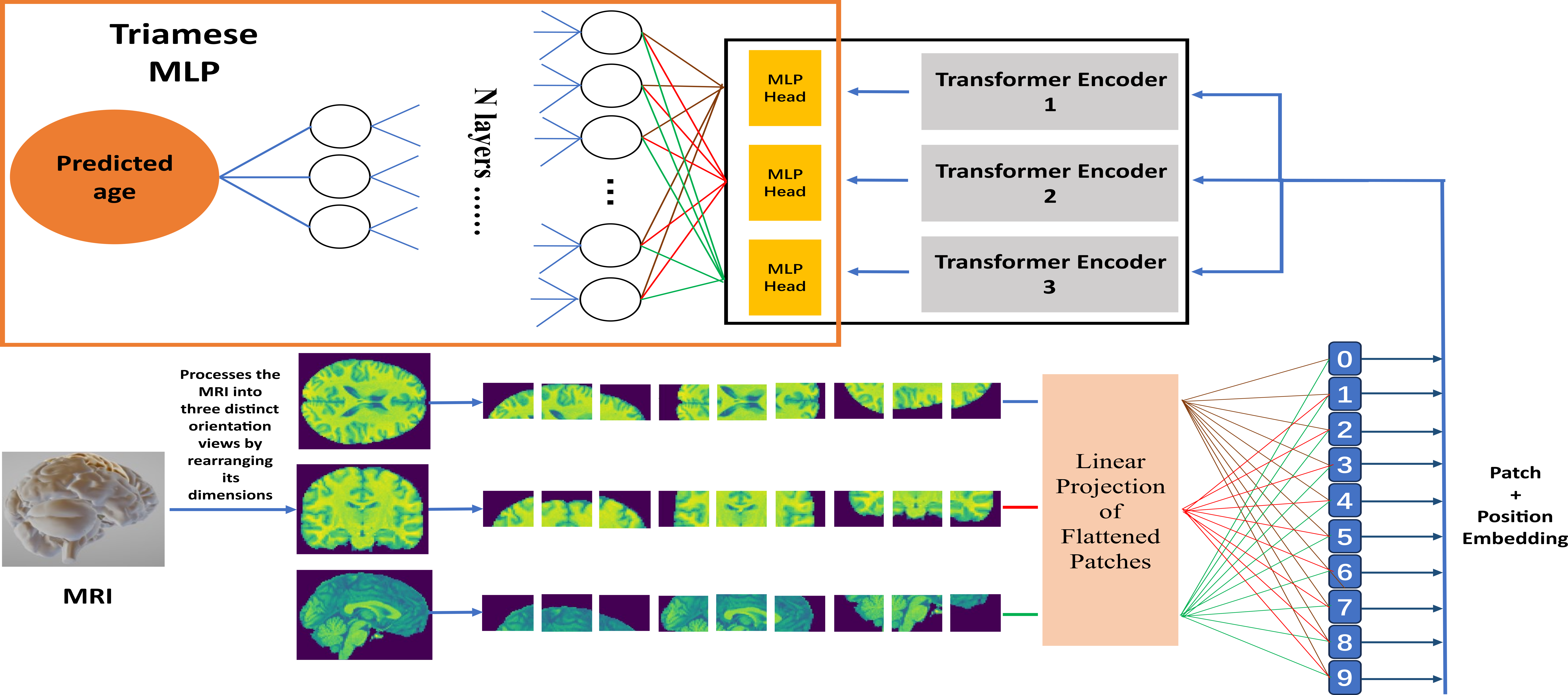
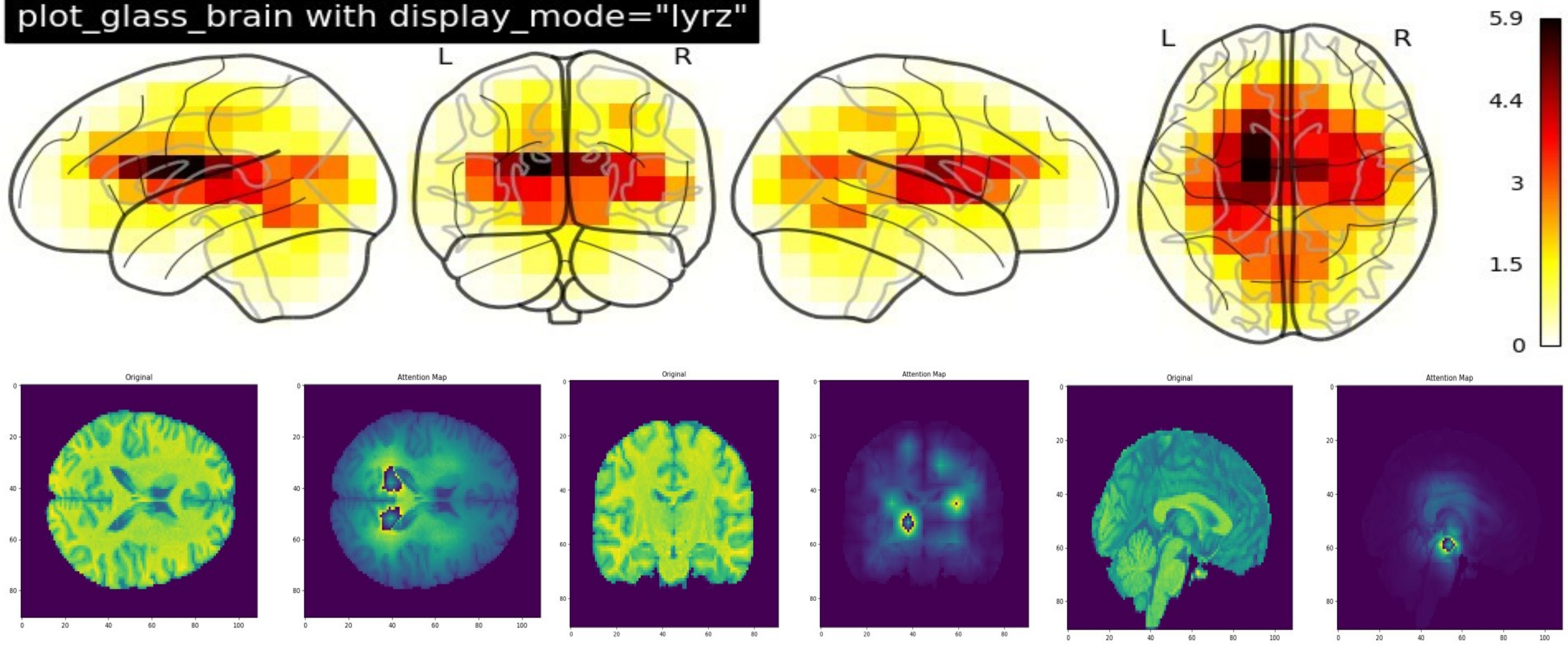
Triamese-ViT: A 3D-Aware Method for Robust Brain Age Estimation from MRIs
Abstract: The integration of machine learning in medicine has significantly improved diagnostic precision, particularly in the interpretation of complex structures like the human brain. Diagnosing challenging conditions such as Alzheimer's disease has prompted the development of brain age estimation techniques. These methods often leverage three-dimensional Magnetic Resonance Imaging (MRI) scans, with recent studies emphasizing the efficacy of 3D convolutional neural networks (CNNs) like 3D ResNet. However, the untapped potential of Vision Transformers (ViTs), known for their accuracy and interpretability, persists in this domain due to limitations in their 3D versions. This paper introduces Triamese-ViT, an innovative adaptation of the ViT model for brain age estimation. Our model uniquely combines ViTs from three different orientations to capture 3D information, significantly enhancing accuracy and interpretability. Tested on a dataset of 1351 MRI scans, Triamese-ViT achieves a Mean Absolute Error (MAE) of 3.84, a 0.9 Spearman correlation coefficient with chronological age, and a -0.29 Spearman correlation coefficient between the brain age gap (BAG) and chronological age, significantly better than previous methods for brian age estimation. A key innovation of Triamese-ViT is its capacity to generate a comprehensive 3D-like attention map, synthesized from 2D attention maps of each orientation-specific ViT. This feature is particularly beneficial for in-depth brain age analysis and disease diagnosis, offering deeper insights into brain health and the mechanisms of age-related neural changes.
- Vision transformer architecture and applications in digital health: a tutorial and survey. Visual Computing for Industry, Biomedicine, and Art, 6(1):14, 2023.
- Explainable artificial intelligence (xai): What we know and what is left to attain trustworthy artificial intelligence. Information Fusion, 99:101805, 2023.
- Review of deep learning: Concepts, cnn architectures, challenges, applications, future directions. Journal of big Data, 8:1–74, 2021.
- Age-net: An mri-based iterative framework for brain biological age estimation. IEEE Transactions on Medical Imaging, 40(7):1778–1791, 2021.
- Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Medical image analysis, 12(1):26–41, 2008.
- Explainable artificial intelligence (xai): Concepts, taxonomies, opportunities and challenges toward responsible ai. Information Fusion, 58:82–115, 2020.
- The association between “brain-age score”(bas) and traditional neuropsychological screening tools in alzheimer’s disease. Brain and Behavior, 8(8):e01020, 2018.
- Predicting brain age with complex networks: From adolescence to adulthood. NeuroImage, 225:117458, 2021.
- Graph transformer geometric learning of brain networks using multimodal mr images for brain age estimation. IEEE Transactions on Medical Imaging, 42(2):456–466, 2022.
- Longbing Cao. Ai in finance: challenges, techniques, and opportunities. ACM Computing Surveys (CSUR), 55(3):1–38, 2022.
- Intelligible models for healthcare: Predicting pneumonia risk and hospital 30-day readmission. In Proceedings of the 21th ACM SIGKDD international conference on knowledge discovery and data mining, pages 1721–1730, 2015.
- Brain age estimation from mri using cascade networks with ranking loss. IEEE Transactions on Medical Imaging, 40(12):3400–3412, 2021.
- Use of machine learning to determine deviance in neuroanatomical maturity associated with future psychosis in youths at clinically high risk. JAMA psychiatry, 75(9):960–968, 2018.
- Predicting age using neuroimaging: innovative brain ageing biomarkers. Trends in neurosciences, 40(12):681–690, 2017.
- Ensemble learning of convolutional neural network, support vector machine, and best linear unbiased predictor for brain age prediction: Aramis contribution to the predictive analytics competition 2019 challenge. Frontiers in Psychiatry, 11:593336, 2020.
- Joint classification and regression for visual tracking with fully convolutional siamese networks. International Journal of Computer Vision, pages 1–17, 2022.
- Opportunities and challenges in explainable artificial intelligence (xai): A survey. arXiv preprint arXiv:2006.11371, 2020.
- Bert: Pre-training of deep bidirectional transformers for language understanding. arXiv preprint arXiv:1810.04805, 2018.
- Towards a rigorous science of interpretable machine learning. arXiv preprint arXiv:1702.08608, 2017.
- An image is worth 16x16 words: Transformers for image recognition at scale. arXiv preprint arXiv:2010.11929, 2020.
- Estimating brain age based on a uniform healthy population with deep learning and structural magnetic resonance imaging. Neurobiology of aging, 91:15–25, 2020.
- Brainage in mild cognitive impaired patients: predicting the conversion to alzheimer’s disease. PloS one, 8(6):e67346, 2013.
- Brain aging in major depressive disorder: results from the enigma major depressive disorder working group. Molecular psychiatry, 26(9):5124–5139, 2021.
- Global-local transformer for brain age estimation. IEEE transactions on medical imaging, 41(1):213–224, 2021.
- Information fusion as an integrative cross-cutting enabler to achieve robust, explainable, and trustworthy medical artificial intelligence. Information Fusion, 79:263–278, 2022.
- Brain age prediction of children using routine brain mr images via deep learning. Frontiers in Neurology, 11:584682, 2020.
- Sqet: Squeeze and excitation transformer for high-accuracy brain age estimation. In 2022 IEEE International Conference on Bioinformatics and Biomedicine (BIBM), pages 1554–1557. IEEE, 2022.
- Age estimation from brain mri images using deep learning. In 2017 IEEE 14th International Symposium on Biomedical Imaging (ISBI 2017), pages 849–852. IEEE, 2017.
- Fsl. Neuroimage, 62(2):782–790, 2012.
- Predicting brain age of healthy adults based on structural mri parcellation using convolutional neural networks. Frontiers in neurology, 10:1346, 2020.
- Transformers in vision: A survey. ACM computing surveys (CSUR), 54(10s):1–41, 2022.
- Accurate brain age prediction using recurrent slice-based networks. In 16th International Symposium on Medical Information Processing and Analysis, pages 11–20. SPIE, 2020.
- Explainability auditing for intelligent systems: a rationale for multi-disciplinary perspectives. In 2021 IEEE 29th international requirements engineering conference workshops (REW), pages 164–168. IEEE, 2021a.
- What do we want from explainable artificial intelligence (xai)?–a stakeholder perspective on xai and a conceptual model guiding interdisciplinary xai research. Artificial Intelligence, 296:103473, 2021b.
- Low-rank atlas image analyses in the presence of pathologies. IEEE transactions on medical imaging, 34(12):2583–2591, 2015.
- Explainable deep learning for personalized age prediction with brain morphology. Frontiers in neuroscience, 15:578, 2021.
- Explainable artificial intelligence (xai) 2.0: A manifesto of open challenges and interdisciplinary research directions. arXiv preprint arXiv:2310.19775, 2023.
- Multimodal biological brain age prediction using magnetic resonance imaging and angiography with the identification of predictive regions. Human brain mapping, 43(8):2554–2566, 2022.
- Improving brain age estimates with deep learning leads to identification of novel genetic factors associated with brain aging. Neurobiology of Aging, 105:199–204, 2021.
- Brain age estimation based on 3d mri images using 3d convolutional neural network. Multimedia Tools and Applications, 79:25051–25065, 2020.
- David Picard. Torch. manual_seed (3407) is all you need: On the influence of random seeds in deep learning architectures for computer vision. arXiv preprint arXiv:2109.08203, 2021.
- A deep ensemble hippocampal cnn model for brain age estimation applied to alzheimer’s diagnosis. Expert Systems with Applications, 195:116622, 2022.
- Age-related midbrain inflammation and senescence in parkinson’s disease. Frontiers in Aging Neuroscience, 14:917797, 2022.
- The impact of the human thalamus on brain-wide information processing. Nature Reviews Neuroscience, pages 1–15, 2023.
- Single-cell sequencing of human midbrain reveals glial activation and a parkinson-specific neuronal state. Brain, 145(3):964–978, 2022.
- Stephen M Smith. Fast robust automated brain extraction. Human brain mapping, 17(3):143–155, 2002.
- Deep learning for brain age estimation: A systematic review. Information Fusion, 2023.
- Roles of the cannabinoid system in the basal ganglia in parkinson’s disease. Frontiers in cellular neuroscience, 16:832854, 2022.
- Accurate brain-age models for routine clinical mri examinations. Neuroimage, 249:118871, 2022.
- Altered functional connectivity of basal ganglia in mild cognitive impairment and alzheimer’s disease. Brain Sciences, 12(11):1555, 2022.
- Siampcf: siamese point regression with coarse-fine classification network for visual tracking. Applied Intelligence, 52(5):4973–4986, 2022.
- Similarity-based pairing improves efficiency of siamese neural networks for regression tasks and uncertainty quantification. Journal of Cheminformatics, 15(1):75, 2023.
- User-centric democratization towards social value aligned medical ai services.
- Robust brain age estimation based on smri via nonlinear age-adaptive ensemble learning. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 30:2146–2156, 2022.
Paper Prompts
Sign up for free to create and run prompts on this paper using GPT-5.
Top Community Prompts
Collections
Sign up for free to add this paper to one or more collections.