- The paper introduces an intricate mathematical framework combining non-local PDEs and physiologically-based pharmacokinetics to model drug resistance in metastatic tumors.
- It employs asymptotic and sensitivity analyses to show how proliferation, migration, and tissue properties influence phenotypic evolution in primary versus metastatic sites.
- Numerical simulations reveal that heterogeneous drug distribution due to tissue-specific vascularization significantly affects long-term treatment outcomes.
The paper presents an intricate mathematical framework to model and analyze the evolutionary dynamics of drug resistance in metastatic tumors treated with chemotherapy. It integrates non-local PDEs for phenotype-structured cancer cell populations with a PK model for drug delivery, focusing on BRAF-mutated melanoma under Dabrafenib treatment.
Model Description and Structure
The study formulates a complex model incorporating two primary components: the evolutionary dynamics of cancer cells and the pharmacokinetics of drug delivery. The evolutionary model employs non-local PDEs to describe the phenotypic distribution of cells in both primary and metastatic sites, with drug resistance being a key phenotypic trait influencing survival and migration.
The pharmacokinetic aspect is captured through a physiologically-based PK model, which simulates the concentration of chemotherapeutic agents across different compartments, including central, peripheral, primary tumor, and metastatic sites.
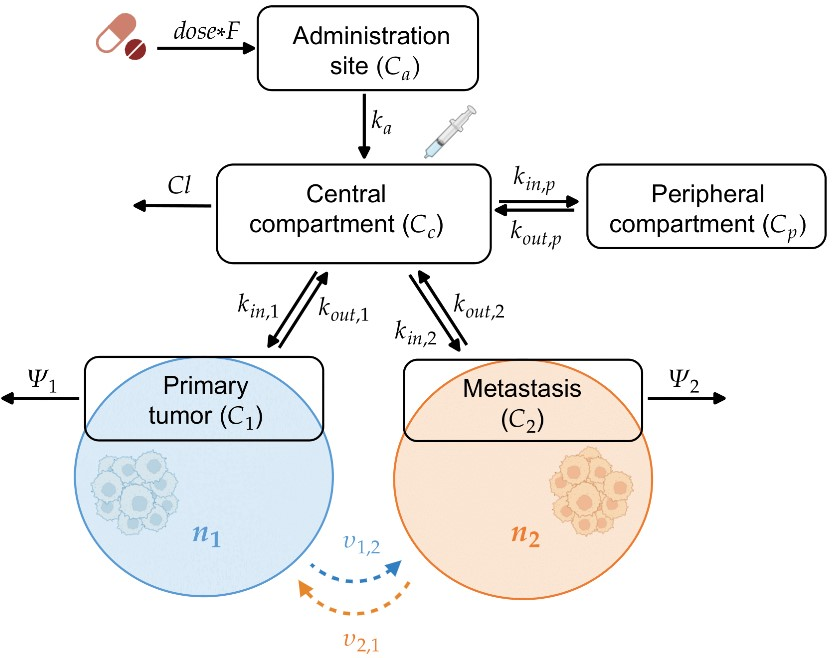
Figure 1: Schematic of the model, illustrating the compartments of the PK model and the interactions between primary and metastatic tumor sites.
Analytical Insights
Asymptotic and Sensitivity Analyses
The paper conducts extensive asymptotic analysis under the assumption of small phenotypic mutation rates, revealing conditions under which tumor populations attain certain phenotypic distributions. Notably, it highlights a tendency for primary tumors to evolve into monomorphic populations when isolated, while interconnected sites tend to exhibit polymorphism.
Global sensitivity analysis (GSA) using the EE and Sobol methods identifies parameters such as proliferation and migration rates as significant influencers of both tumor mass and phenotypic state at equilibrium. These analyses elucidate the complex interplay between drug dynamics and tumor evolution, emphasizing the adaptive nature of metastatic tumors.
Numerical Simulations and Real-World Implications
Simulations grounded in the baseline scenario reveal that differences in tissue-to-plasma partition coefficients can lead to heterogeneous drug distributions between tumor sites, driving distinct evolutionary outcomes. The model predicts that metastatic sites with lower vascularization exhibit slower drug accumulation, fostering phenotypic diversity with a subset of resistant cells.
An important finding concerns the dynamics of treatment delivery. Intravenous injection ensures more stable drug concentration compared to oral administration, which exhibits periodic pharmacokinetics. However, the study highlights that long-term therapeutic outcomes depend more on average in situ drug concentrations than temporal fluctuations.
Conclusion and Prospective Directions
The research offers profound insights into the evolutionary dynamics driving drug resistance in metastatic tumors. By simulating different scenarios, the model suggests that site-specific properties, such as vascularization and migration rates, critically influence treatment efficacy and cancer cell adaptation.
Future work could expand this framework to include multi-drug protocols and refine migration models to better capture the stochastic nature of metastatic spread. Integrating patient-specific data could enhance the model's applicability in personalized therapy planning, paving the way for precision oncology.
