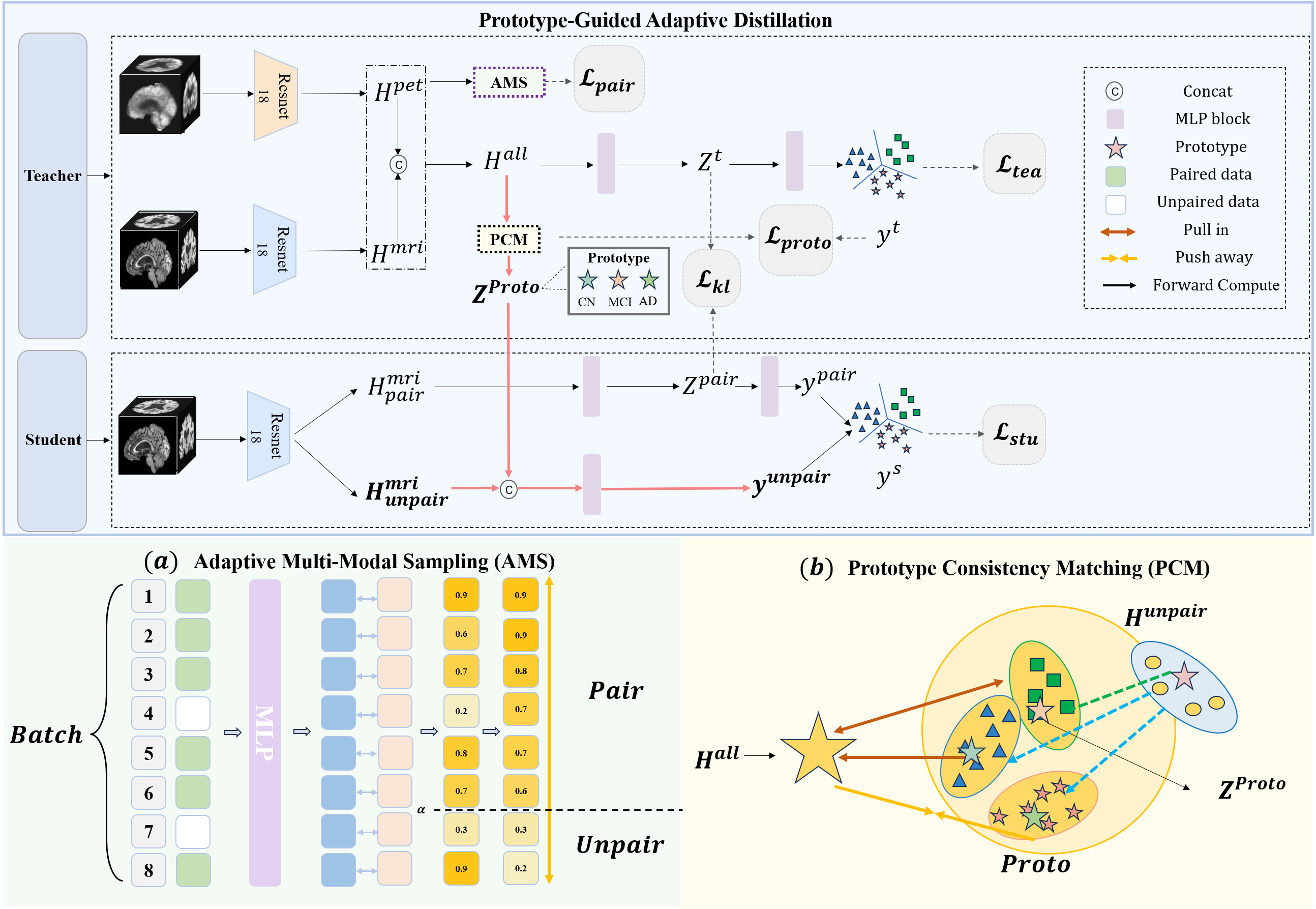
- The paper introduces PGAD, a novel framework using prototype matching and adaptive sampling to boost AD diagnostic accuracy under missing modality conditions.
- It integrates Prototype Consistency Matching (PCM) and Adaptive Multi-Modal Sampling (AMS) to align features from MRI and PET data, ensuring robust multi-modal learning.
- Experimental results on ADNI demonstrate superior performance with a mean MCC of 85.1 and AUC of 96.3%, highlighting its clinical applicability.
PGAD: Prototype-Guided Adaptive Distillation for Multi-Modal Learning in AD Diagnosis
Introduction
The challenges of diagnosing Alzheimer's Disease (AD) are compounded by missing data modalities, particularly in multi-modal neuroimaging. PET imaging, despite its advantages, is often missing due to high costs, yielding incomplete datasets. The paper introduces the Prototype-Guided Adaptive Distillation (PGAD) framework, which innovatively tackles missing modality issues in AD diagnosis by leveraging prototype matching and adaptive sampling strategies. PGAD is validated on the ADNI dataset under varying missing rates, revealing its superior performance over existing techniques.
Methodology
The PGAD framework is designed to integrate incomplete multi-modal data efficiently, improving AD diagnosis accuracy even under significant data incompleteness. It comprises two core components:
Experimental Results
The PGAD framework's efficacy was evaluated on the ADNI dataset across different modality missing rates (20%, 50%, 70%). Its performance was benchmarked against state-of-the-art methods, exhibiting superior outcomes in AD classification and MCI conversion prediction. Notably, PGAD achieved a mean MCC of 85.1 and an AUC of 96.3% for AD vs. CN classification, outperforming alternatives such as DFTD and MDT-Student in sensitivity and specificity, crucial for clinical diagnosis.
Ablation Studies
A comprehensive ablation study delineated the contributions of PGAD's individual components. Removing PCM or AMS resulted in significant performance degradation, underscoring their role in maintaining feature alignment and model robustness. The studies confirmed that PCM effectively preserves class-discriminative features, while AMS ensures stable learning through optimal data balance in training.
Visualization Analysis
t-SNE and Grad-CAM visualizations were employed to qualitatively validate PGAD's feature learning and interpretability. t-SNE plots highlighted substantial improvements in feature clustering and separation with PGAD compared to baseline configurations (Figure 2). Grad-CAM visualizations revealed the model's focus on clinically relevant regions like the hippocampus, validating its biological plausibility and potential for trustworthy diagnostic support.
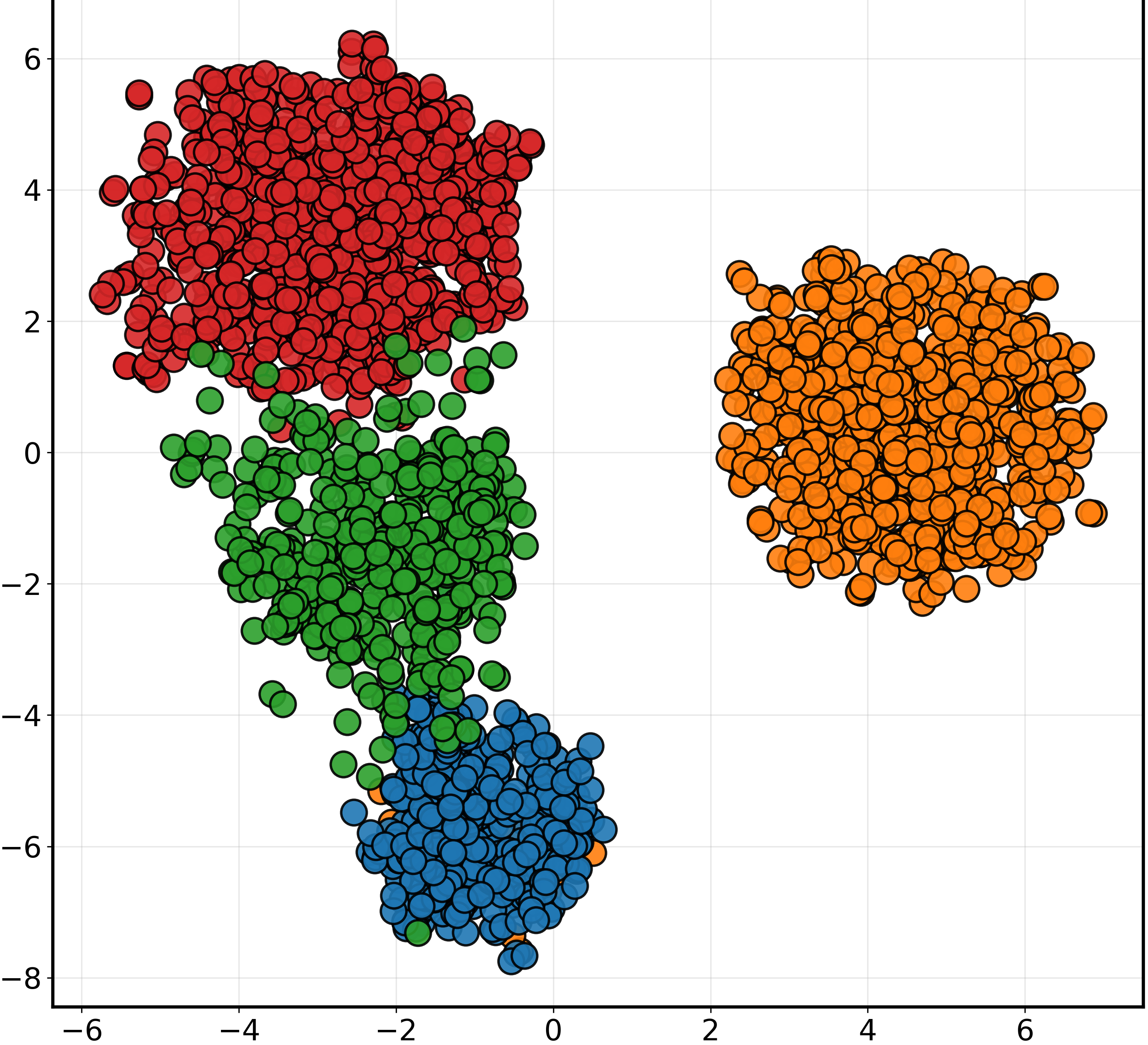
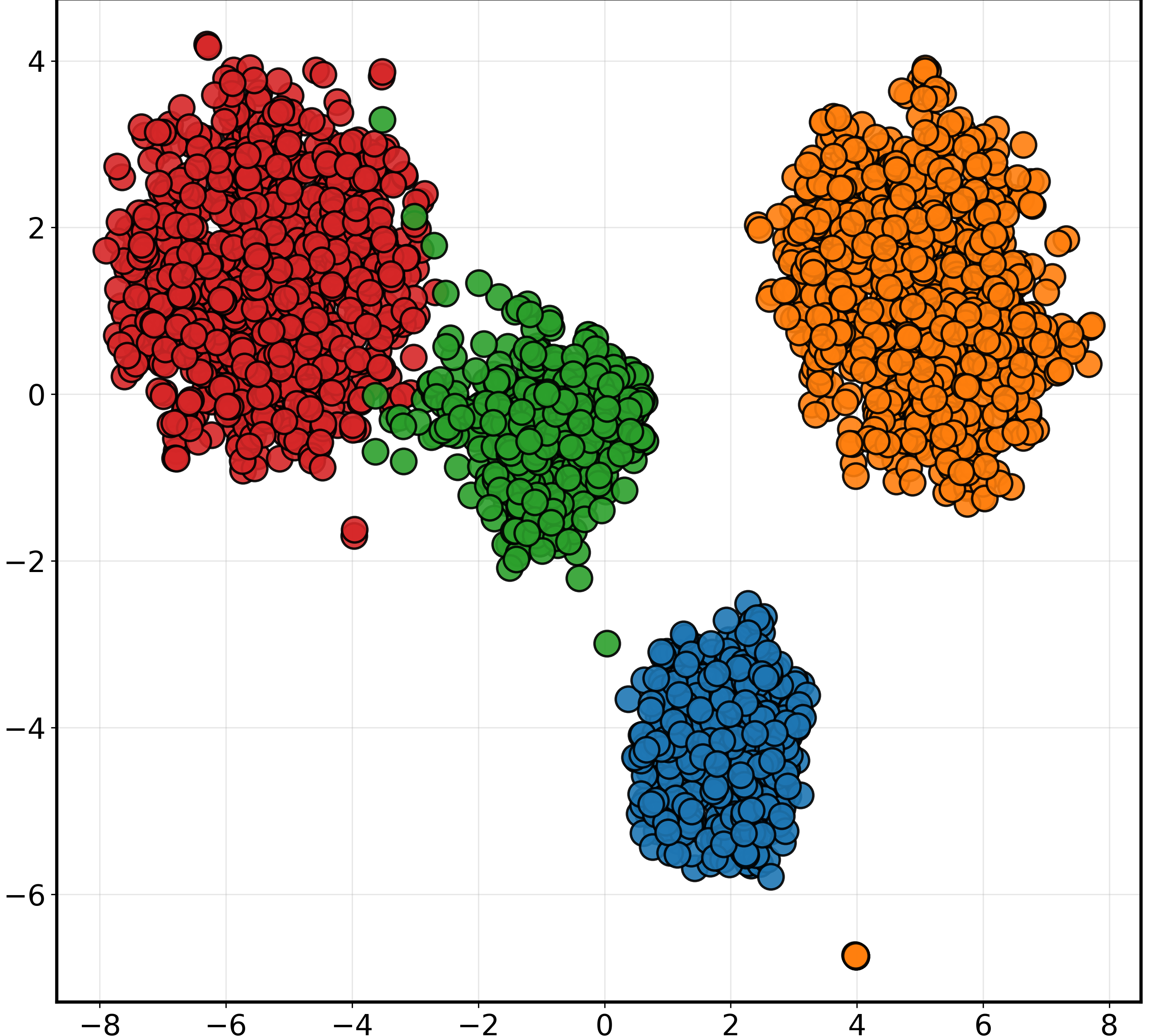
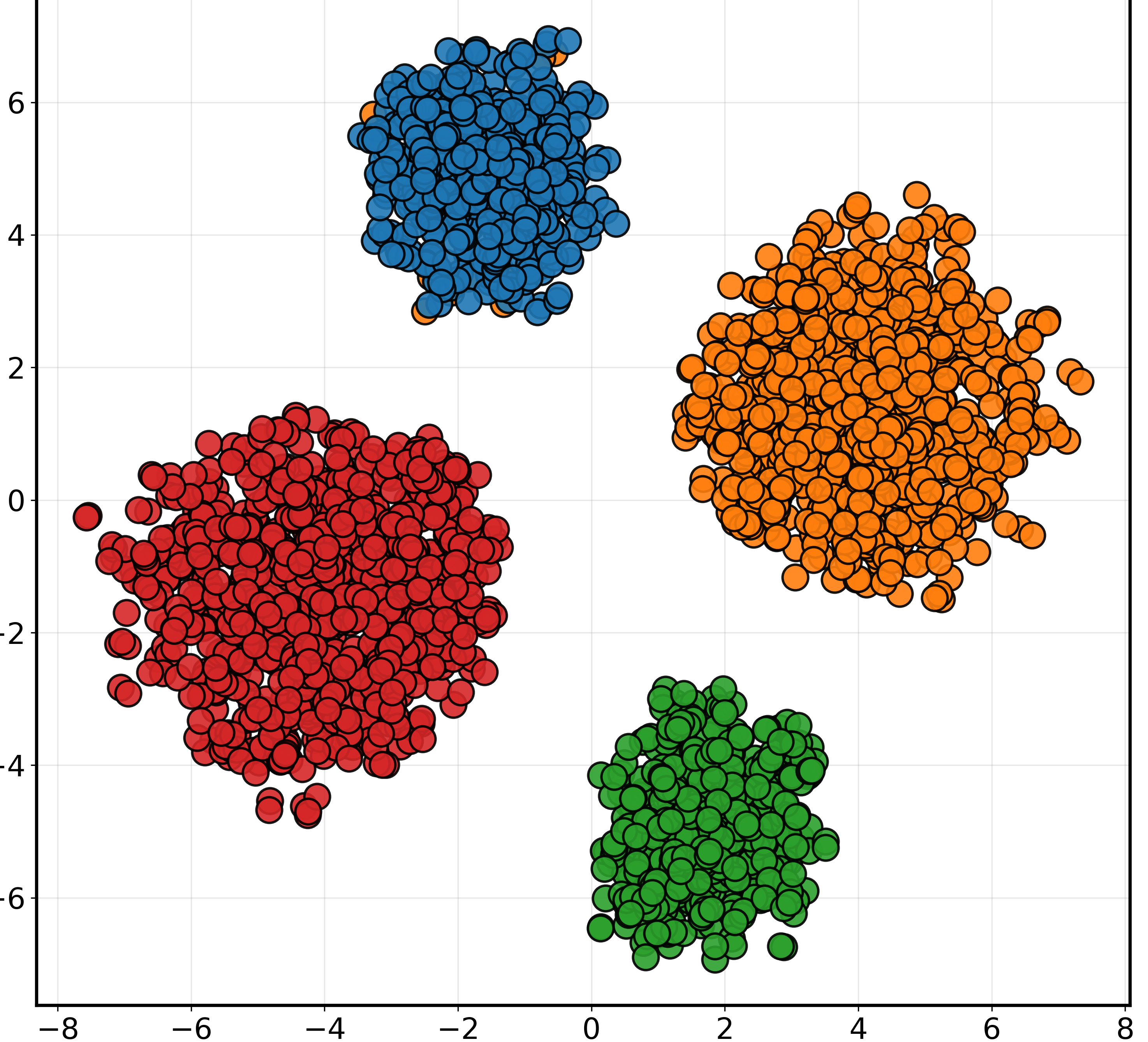

Figure 2: t-SNE visualization of MRI feature embeddings under different training configurations (50% missing modality).
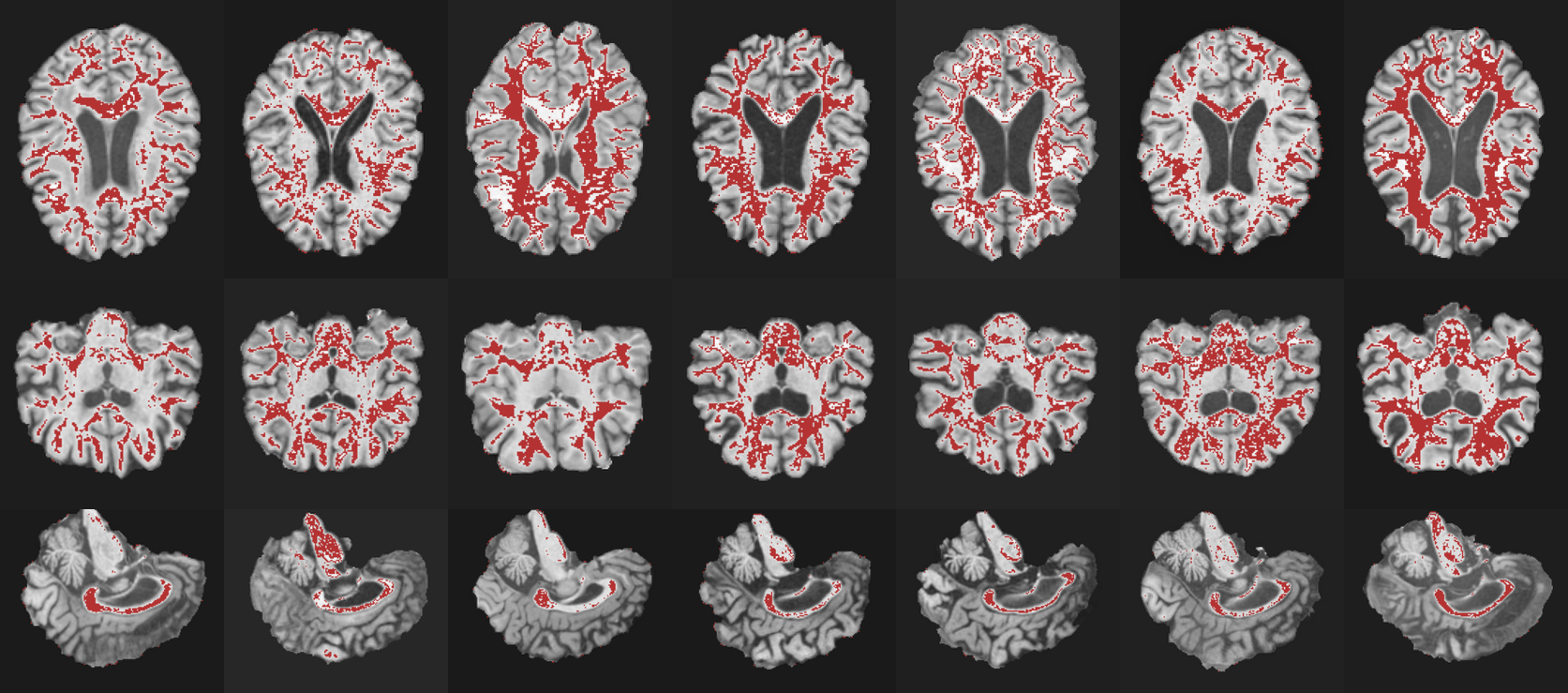
Figure 3: Grad-CAM visualization of the model's attention regions on MRI slices. The red-highlighted areas indicate regions most relevant to AD classification in axial, coronal, and sagittal views.
Conclusion
The PGAD framework represents a significant advancement in multi-modal learning for AD diagnosis under missing data conditions. By integrating novel prototype-guided learning and adaptive sampling strategies, PGAD effectively mitigates the challenges of incomplete data, enhancing diagnostic accuracy and model robustness. This study paves the way for future research in applying prototype-guided adaptive distillation to more complex, multi-center clinical datasets. The framework's capability to operate under high missing modality rates highlights its practical applicability and potential for widespread adoption in real-world clinical settings.