- The paper demonstrates how resource allocation theory explains metabolic trade-offs, showing that cancer cells optimize both energy production and biosynthesis.
- The paper compares microbial overflow metabolism with the cancer-associated glycolytic shift, highlighting similarities in energy efficiency and regulatory mechanisms.
- The paper illustrates how insights from microbial models can inform strategies to target cancer’s unique metabolic landscape.
Introduction
The cellular metabolism of cancer cells is characterized by a preference for glycolytic processes even in the presence of oxygen, a phenomenon traditionally referred to as the Warburg effect. Historically, this shift in metabolic preference has been observed in cancer cells, where the adoption of aerobic glycolysis over oxidative phosphorylation is thought to provide a growth advantage. The paper "Balancing the Cellular Budget: Lessons in Metabolism from Microbes to Cancer" (2506.20776) analyzes this metabolic choice not merely as a simplistic growth prioritization but through a multifaceted lens of optimization principles observed in both cancer and microbial systems.
Cancer cells exhibit a tendency to switch metabolic pathways from oxidative phosphorylation to glycolysis, which allows for rapid ATP production albeit at lower efficiency. This metabolic flexibility is not solely about optimizing growth rates but also involves balancing biosynthetic demands against energetic costs. The metabolic phenotype of cancer emerges from complex optimization, where resource allocation theory provides a basis for understanding these choices. The metabolic landscape in cancers is influenced by genetic mutations, microenvironmental conditions, and various external nutrient availabilities.
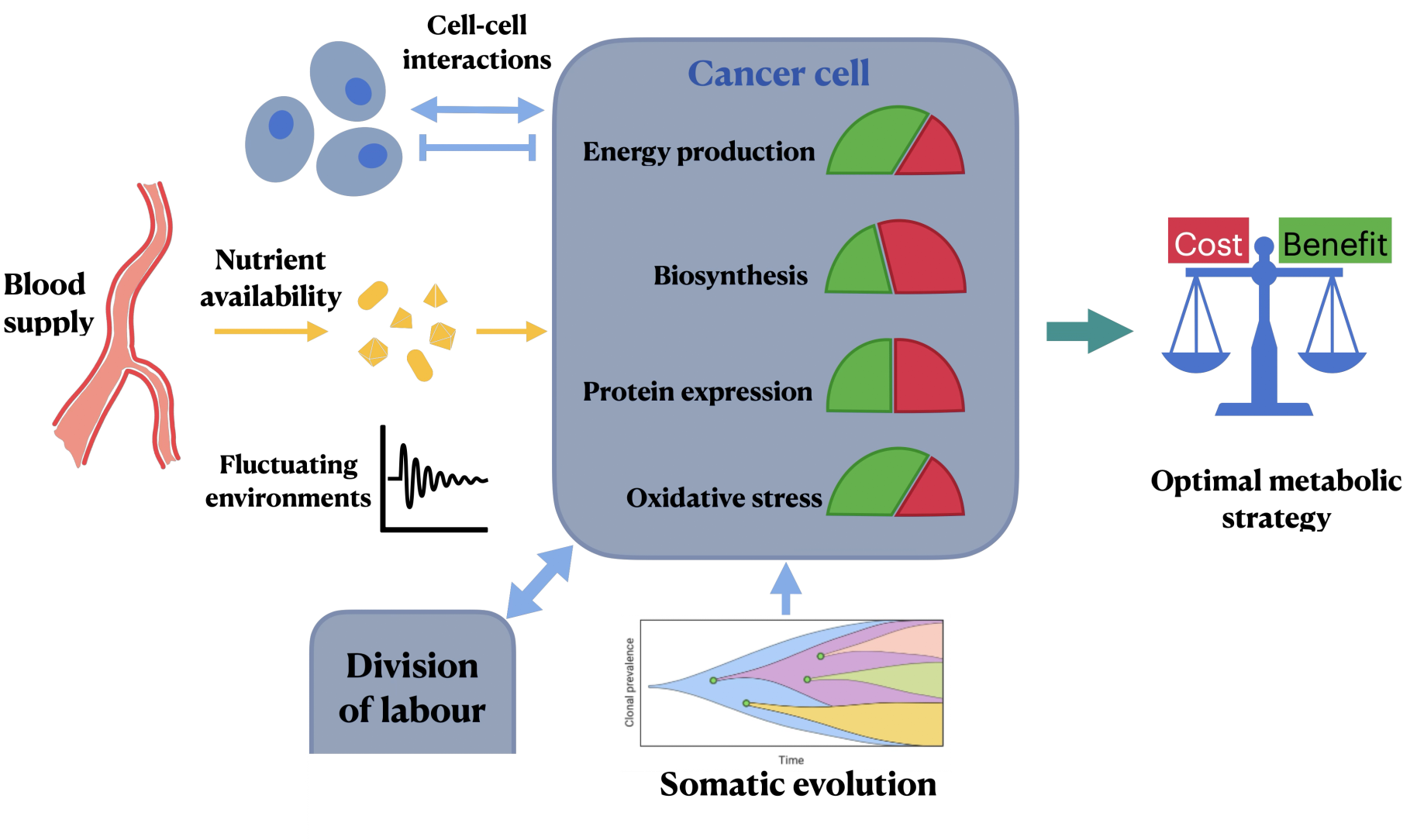
Figure 1: Resource allocation as a rational basis for cancer metabolism.
Analogous to cancer cells, microbial systems such as bacteria showcase overflow metabolism—favoring glycolysis under nutrient-abundant conditions. Despite their reliance on different molecular pathways, both microbes and cancer cells manage metabolic strategies driven by a trade-off between protein expression costs, nutrient usage efficiency, and the energetic demands of rapid reproduction. The concept of resource allocation has been pivotal in providing insights into the microbial approach to metabolic optimization, highlighting the potential to apply similar frameworks to unveil the underlying principles governing cancer metabolism.
Application of Resource Allocation Theory
Resource allocation theory elucidates the choice between metabolic states by dissecting the interplay of bioenergetics, nutrient availability, and cellular machinery costs in microbial systems. Cancer cells, akin to microbes, optimize metabolic states based on environmental contexts and intrinsic cellular demands. This approach not only rationalizes the presence of hybrid metabolic states within cancer cells, where both pathways coexist, but also provides a framework for exploring cellular metabolic strategies as emergent properties governed by cost-benefit analyses in varying conditions.
While the overlap with microbial metabolism is noteworthy, cancer metabolism is further complicated by somatic mutations that irrevocably alter metabolic network topologies. The external biochemical environment in which cancer cells thrive is vastly different from typical culture conditions, adding layers of complexity to their metabolic regulation. Furthermore, fluctuating nutrient supplies in tumor microenvironments necessitate a flexible metabolic strategy, potentially paving the way for new therapeutic avenues to target cancer metabolism effectively.
Conclusion
The comparative review provided in "Balancing the Cellular Budget: Lessons in Metabolism from Microbes to Cancer" posits the resource allocation framework as a viable approach to understanding cancer metabolism. Although the mechanistic intricacies of cancer metabolism exhibit unique traits beyond normal cellular physiology, leveraging this integrative framework could yield novel insights into efficient metabolic strategies adopted by cancer cells. By encapsulating diverse observations across cancers, this perspective holds promise in reconciling disparate metabolic states within a coherent theoretical structure, thereby advancing the understanding of cancer metabolic phenotypes and enhancing therapeutic targeting strategies.
