- The paper presents a first-principles study showing that SnSiGeN4 has a direct band gap and excellent photocatalytic activity for OER, ORR, and HER.
- It employs DFT with various exchange-correlation functionals to validate structural stability and favorable electronic, vibrational, and optical properties.
- The study finds near-zero Gibbs free energy changes at specific active sites, indicating superior catalytic performance, potentially outperforming platinum.
Insights into the OER, ORR, and HER Activity of a New MXene-Family SnSiGeN₄ Photocatalyst for Water Splitting: A First-Principles Study
Introduction
The study explores the photocatalytic properties of a novel SnSiGeN₄ MXene-family monolayer, aiming to provide a cost-effective alternative to platinum for water splitting reactions. Using first-principles calculations, the paper examines the electronic, vibrational, and optical characteristics of SnSiGeN₄, highlighting its potential as a sustainable catalyst for the Oxygen Evolution Reaction (OER), Oxygen Reduction Reaction (ORR), and Hydrogen Evolution Reaction (HER).
Computational Methods and Structural Properties
The research employed density functional theory (DFT) utilizing the CRYSTAL17 ab initio code with various exchange-correlation functionals to model the equilibrium geometries and electronic properties of SnSiGeN₄. The optimized monolayer structure (Figure 1) demonstrates stability with hexagonal lattice configurations.
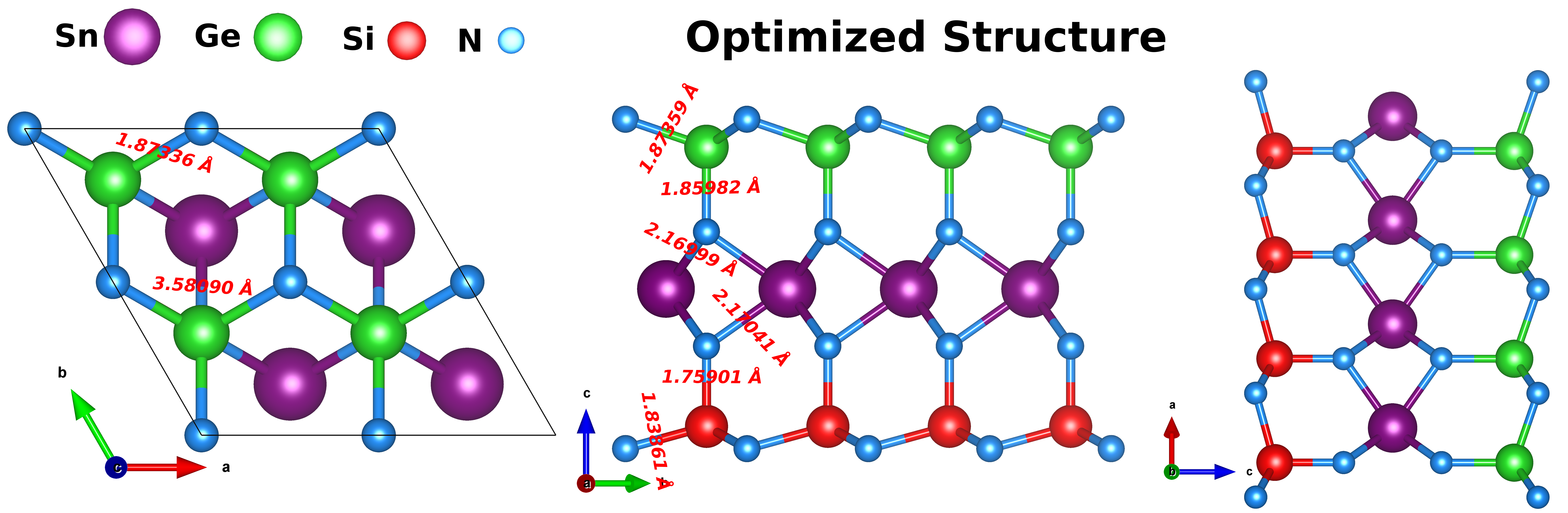
Figure 1: Top and side views of the optimized SnSiGeN₄ monolayer. The structure belongs to the hexagonal lattice system with space group P1 (No. 1). Atoms are labelled, and red-colored lines represent the calculated bond lengths between adjacent atoms.
Electronic and Optical Properties
SnSiGeN₄ exhibits a direct band gap, the value of which depends on the functional used. Hybrid functionals like HSE06 predict a gap of around 2.92 eV, suitable for visible-light-driven photocatalysis. Band edge alignment diagrams (Figure 2) indicate feasibility for water splitting across a wide pH range.
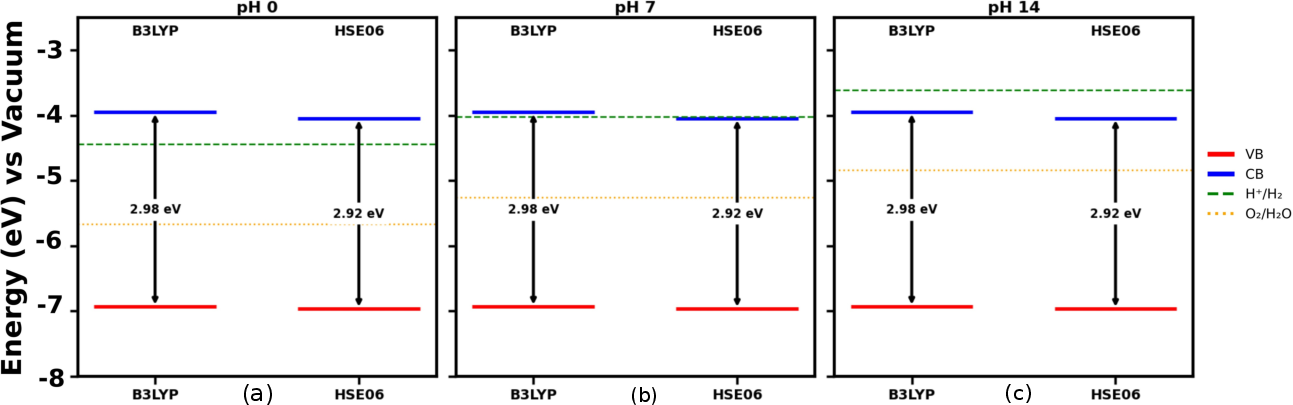
Figure 2: Schematic Representation of Band Edge Allignment at different pH.
The projected density of states (PDOS) analyses (Figure 3) reveal strong contributions from N and Si orbitals at the band edges, favoring charge separation and transport, which are crucial for photocatalytic activity.
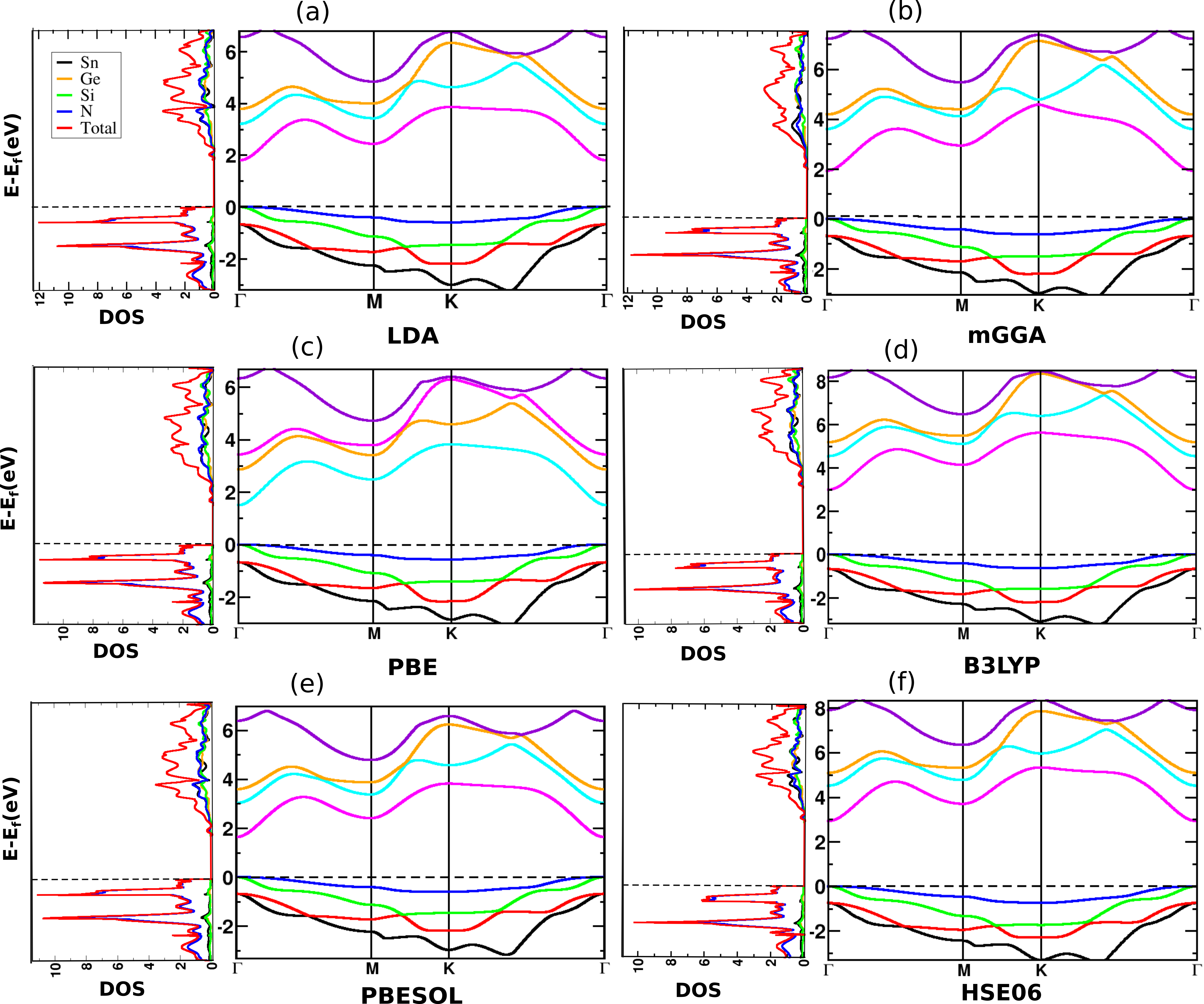
Figure 3: Projected density of states (PDOS) and electronic band structures of SnSiGeN₄ monolayer calculated using different exchange-correlation functionals.
The monolayer's strong absorption in the UV-visible spectrum (Figure 4) further enhances its potential as a photocatalyst by promoting efficient photon capture and charge carrier generation.
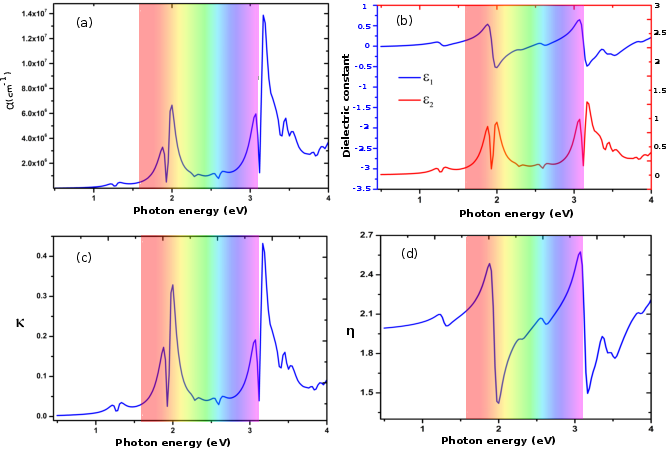
Figure 4: (a) absorption coefficient, (b) Dielectric constant, (c) extinction function, (d) refractive index.
Vibrational Properties
Vibrational analysis using IR and Raman spectra (Figures 5 and 6) reveals stability and the presence of catalytically active modes. Specific modes show potential to enhance the performance of catalytic sites, facilitating intermediate adsorption crucial for catalysis.
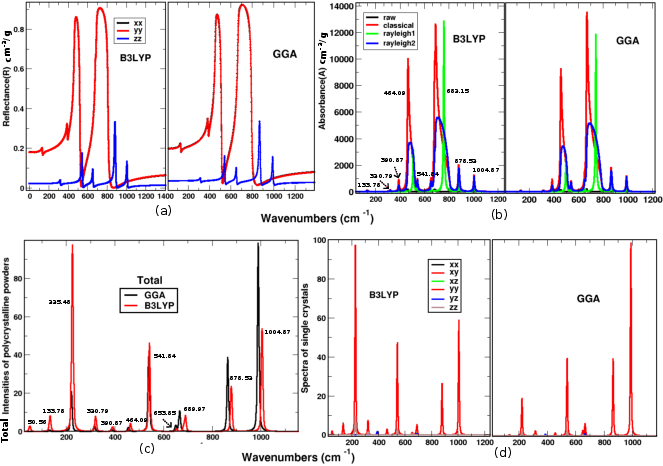
Figure 5: Vibrational IR and Raman spectra of the SnSiGeN₄ monolayer calculated with B3LYP and GGA functionals. The numerical label denotes the respective frequency mode.
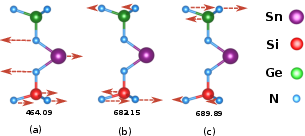
Figure 6: Representative IR active vibrational mode in SnGeSiN₄which enhance the activation of catalytic sites by facilitating intermediate adsorption and stabilization.
Catalytic Activity
OER and ORR Activity
The Gibbs free energy diagrams (Figures 7 and 8) for OER and ORR on SnSiGeN₄ show low overpotentials, indicating excellent catalytic efficiency. For OER, site V demonstrates the lowest overpotential (0.48 V), outperforming traditional IrO₂.
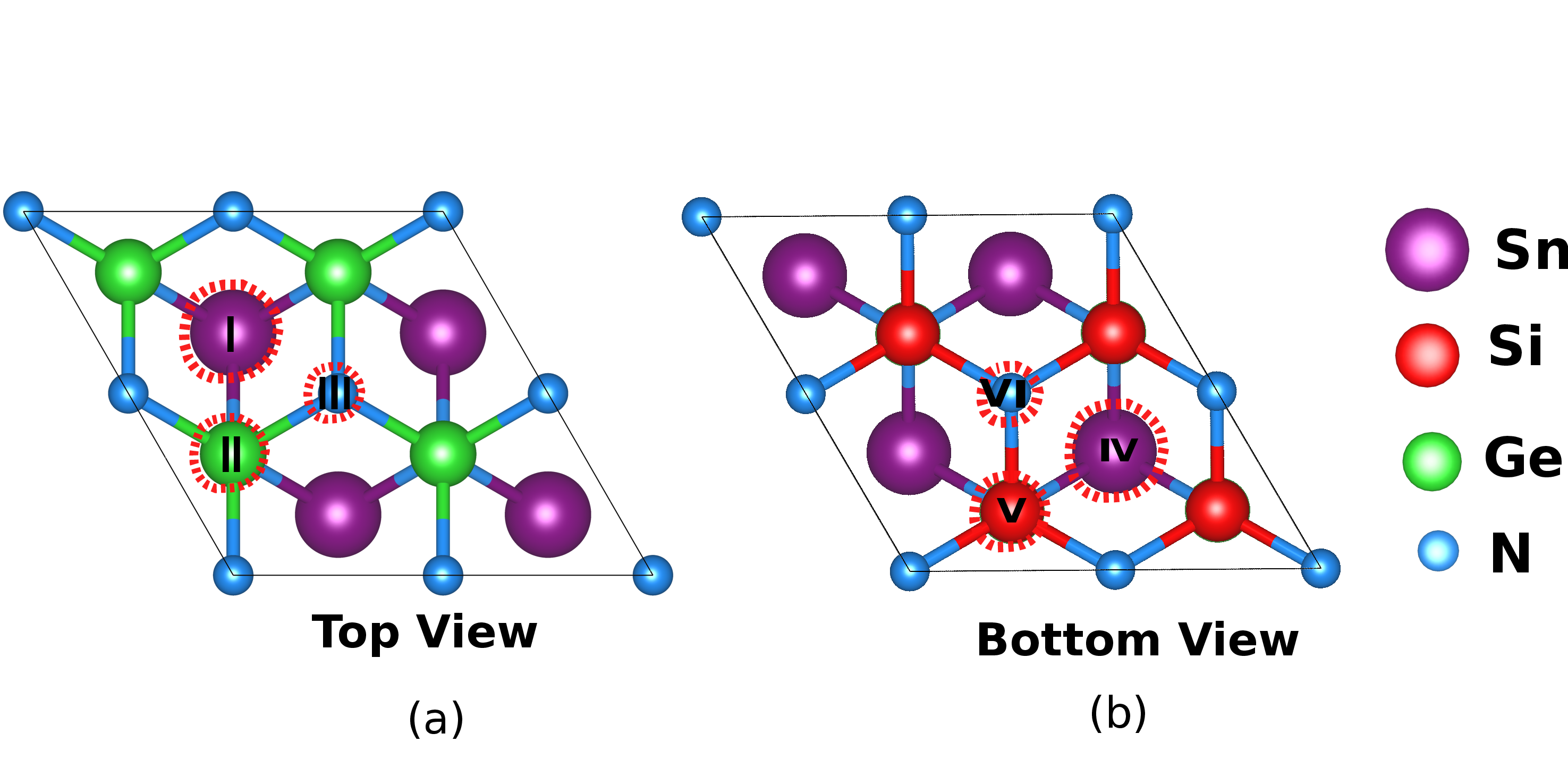
Figure 7: Different possible active sites for OER, ORR and HER reactions.
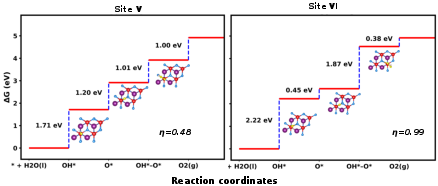
Figure 8: Gibbs free energy diagram of the OER pathway on the surface of the 2D monolayer SnSiGeN₄ with optimized structures.
HER Activity
Site-specific analyses (Figure 9) demonstrate favorable adsorption and release of hydrogen, with specific sites surpassing Pt in activity. The optimized sites show close-to-zero Gibbs free energy change, reinforcing their enhanced catalytic performance.
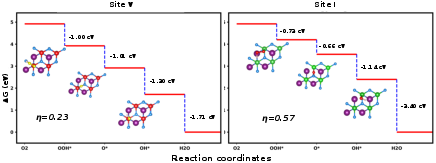
Figure 9: Gibbs free energy diagram of the ORR pathway on the surface of the 2D monolayer SnSiGeN₄ with optimized structures.
Conclusion
The study positions SnSiGeN₄ as a viable, efficient photocatalyst with significant potential to replace costly noble metals in water-splitting applications. Its tunable electronic structure, combined with stability and low overpotentials, paves the way for practical energy solutions in hydrogen production. Future work could experimentally validate these theoretical predictions and explore scalability in real-world applications.








