- The paper quantifies the physical impact of urease-driven enzyme activity on protein liquid-liquid phase separation using a chemostatic microfluidic system.
- It demonstrates that the active bath significantly increases droplet size, number density, and area fraction—with droplets nearly doubling or tripling compared to controls.
- Coarse-grained simulations and experiments reveal that enzyme activity acts as an effective temperature, shifting the LLPS phase boundary and promoting protein condensation.
Enzyme-Driven Active Baths Modulate Protein Liquid-Liquid Phase Separation
Introduction
This study rigorously investigates the physical impact of enzymatic activity on protein liquid-liquid phase separation (LLPS), focusing on the interplay between a model phase-separating protein, truncated ubiquilin-2 (UBQLN2-450C), and an active bath of urease enzymes. The work isolates the physical effects of enzyme-driven activity from chemical changes by employing a chemostatic microfluidic device, enabling precise control over substrate and product concentrations. The central claim is that the active bath enhances protein condensation, acting as an effective temperature and shifting the phase behavior of the system. This is substantiated by both experimental quantification and coarse-grained molecular dynamics simulations.
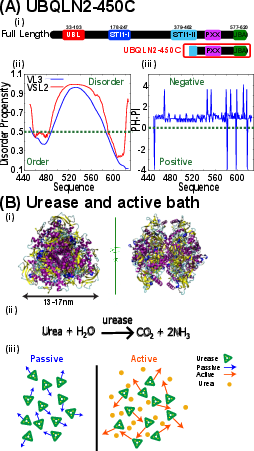
Figure 1: Schematic of the model systems: truncated UBQLN2-450C as the phase-separating protein and urease as the active bath enzyme, including charge and disorder propensity.
Experimental System and Chemostatic Control
The experimental design utilizes a microfluidic chamber with semipermeable membrane walls, allowing for the continuous supply of urea and removal of reaction products, thereby maintaining a chemostatic environment. The central lane contains the protein system, while the outer lanes facilitate substrate delivery and product removal. Surface passivation with Pluronic F127 prevents protein adhesion, ensuring accurate droplet quantification.
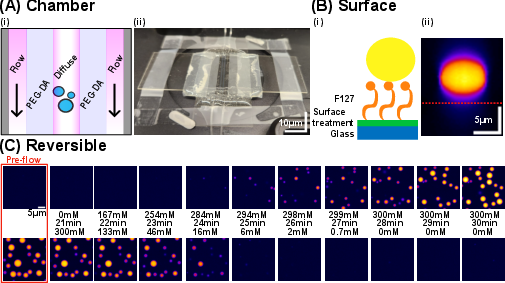
Figure 2: Microfluidic device for chemostatic control, including chamber schematic, surface passivation, and reversible control of UBQLN2-450C phase transition via NaCl.
Three conditions are compared: (1) active bath (urease + urea), (2) no-activity control (urease without urea), and (3) product control (urease + reaction products). This design ensures that observed effects are attributable to physical activity rather than chemical changes.
Quantitative Effects of Enzyme Activity on Protein Condensation
Droplet Size, Density, and Area Fraction
Spinning disc confocal microscopy and automated image analysis reveal that the active bath condition produces significantly larger and more numerous UBQLN2-450C droplets compared to controls. The cumulative distribution functions (CDFs) of droplet areas are statistically distinct (KS test, p<0.01), with the active bath yielding a mean droplet area approximately twice that of the no-activity control and three times that of the product control. Droplet density is also enhanced, with the active bath showing a twofold increase over the no-activity control.
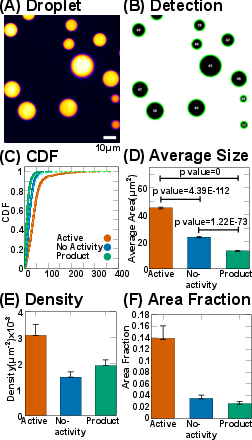
Figure 3: Quantitative analysis of droplet size, number density, and area fraction for active bath, no activity, and product control conditions.
The area fraction occupied by droplets in the active bath is four to five times higher than in controls, indicating a substantial shift in the phase diagram toward increased condensation.
Partition Coefficient and Bulk Protein Quantification
Fluorescence intensity measurements within droplets and the surrounding dilute phase yield partition coefficients (PC) that are markedly higher in the active bath (PC ≈ 30) compared to controls (PC ≈ 13–15). Bulk spectrophotometric analysis of the dilute phase after centrifugation confirms that the active bath drives more protein into the condensed phase, with the dilute phase concentration in the active bath being 3–6 times lower than in controls.
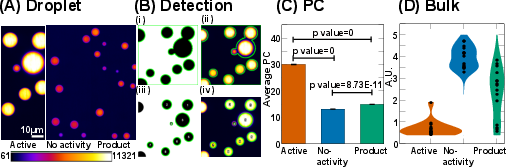
Figure 4: Partition coefficient quantification and bulk measurement of UBQLN2-450C in condensed and dilute phases across conditions.
Enzyme Partitioning and Droplet Material Properties
Partitioning of urease into droplets is modest (PC ≈ 1.3–1.37) and only slightly enhanced by activity, contrasting with previous reports of strong enzyme partitioning in other systems. Fluorescence recovery after photobleaching (FRAP) experiments show a small but significant decrease in recovery time and mobile fraction in the active bath, indicating increased protein mobility and interaction strength within droplets.
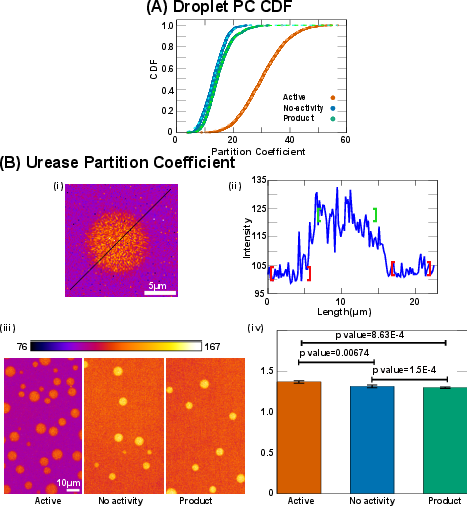
Figure 5: Partition coefficient distributions for UBQLN2-450C and urease, with intensity profiles and microscopy images.
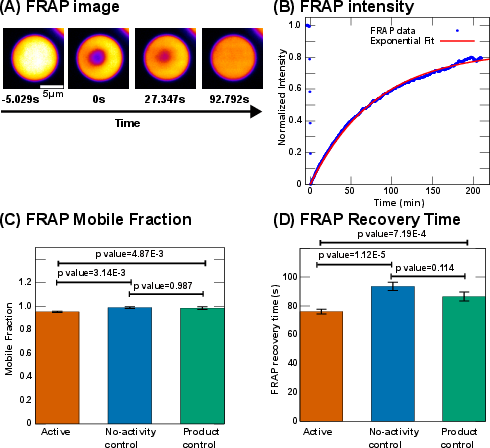
Figure 6: FRAP analysis of mobile fraction and recovery time for UBQLN2-450C droplets under different bath conditions.
Coarse-Grained Simulations: Mechanistic Insights
Sticker-spacer polymer simulations in LAMMPS model the protein as alternating A/B stickers connected by spacers, with bath particles representing urease. Increasing the bath activity (Peclét number, Pe) enhances droplet size and polymer partitioning, consistent with experimental results. The polymer partition coefficient increases by 1.5-fold in the active bath, while bath particle partitioning remains low. At high binding affinity, polymers in the dense phase adopt more extended conformations, accommodating bath particles and promoting cluster growth.
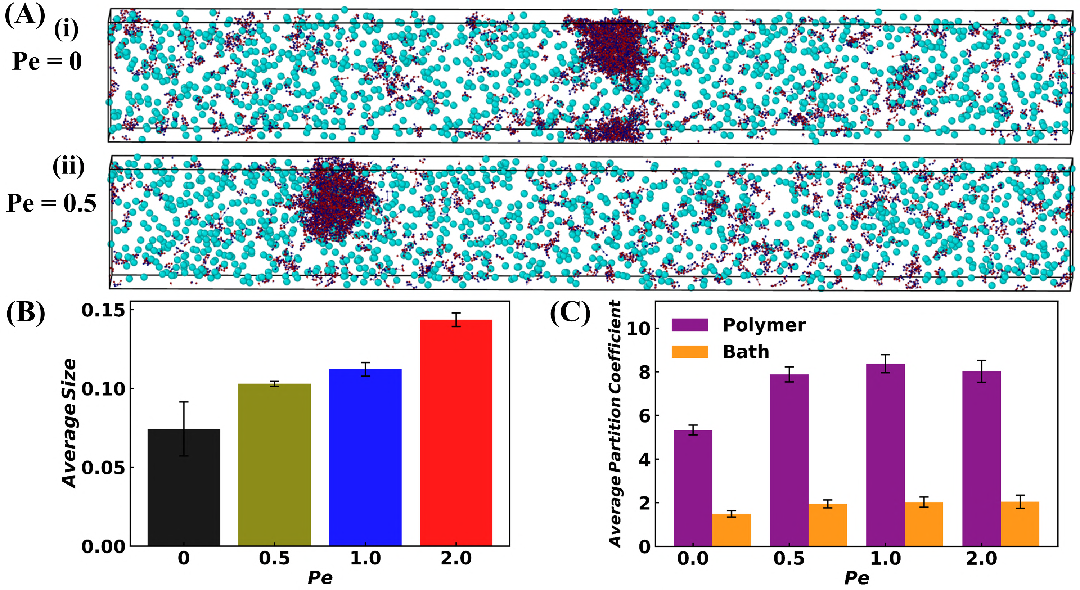
Figure 7: Simulation snapshots and quantitative analysis of droplet size and partition coefficients as a function of bath activity.
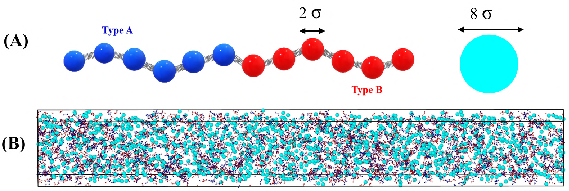
Figure 8: Schematic and simulation snapshot of sticker-spacer polymers and bath particles in an active bath.
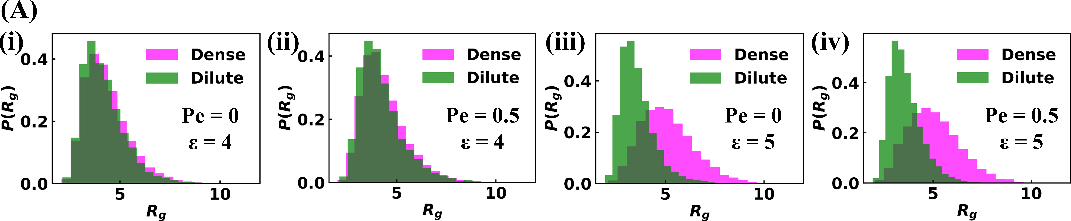
Figure 9: Distribution of polymer radius of gyration (Rg) distinguishing dilute and droplet-like states at different binding affinities and bath activities.
Discussion and Implications
The data robustly support the assertion that enzymatic activity in the bath acts as an effective temperature, shifting the LLPS phase boundary and driving more protein into the condensed phase. This effect is physical rather than chemical, as demonstrated by the chemostatic control and comparison to product controls. The findings extend active matter theory to the nanoscale, showing that enzyme-driven fluctuations can modulate protein organization and material properties.
Contrary to previous studies where active baths destroyed condensates via chemical gradients or mechanical stretching, this work demonstrates that physical activity can enhance condensation. Theoretical models suggest that activity not only increases effective temperature but also renormalizes interaction strengths, a dual effect observed in both experiment and simulation.
These results have broad implications for understanding intracellular organization, where enzymatic activity may serve as a physical regulator of biomolecular condensates. The approach also suggests strategies for engineering bio-inspired active materials with tunable hierarchical assembly.
Conclusion
This study provides quantitative evidence that an enzyme-driven active bath enhances protein condensation by shifting the LLPS phase boundary, increasing droplet size, density, and protein concentration in the condensed phase. The effect is physical, not chemical, and is corroborated by coarse-grained simulations. These findings advance the understanding of active matter in biological systems and open avenues for the design of tunable active materials.








