- The paper presents a consensus-driven hybrid framework that enhances single-trial P300 classification by integrating data-driven ROI selection with cross-subject functional connectivity.
- It employs eLORETA-based source localization and phase synchronization metrics (PLI, wPLI, dPLI) to achieve up to 81.57% accuracy in the 1–15 Hz band.
- The approach offers actionable insights for neurodegenerative biomarker detection and portable EEG-based BCI applications by isolating neurophysiologically relevant hubs.
Data-Driven ROI Selection for Enhanced Single-Trial P300 Classification in EEG-Based BCIs
Introduction
The paper presents a consensus-driven hybrid framework for single-trial P300 detection in EEG-based BCIs, leveraging data-driven region-of-interest (ROI) selection via eLORETA source localization and cross-subject functional connectivity. The approach addresses the spatial specificity limitations inherent in non-invasive EEG, particularly for detecting P300 potentials—key neurophysiological markers for BCI control and neurodegenerative disease diagnostics. By integrating spatially stable ROIs with dynamic temporal agreement, the framework systematically outperforms conventional whole-brain methods, achieving up to 81.57% accuracy in the 1–15 Hz band.
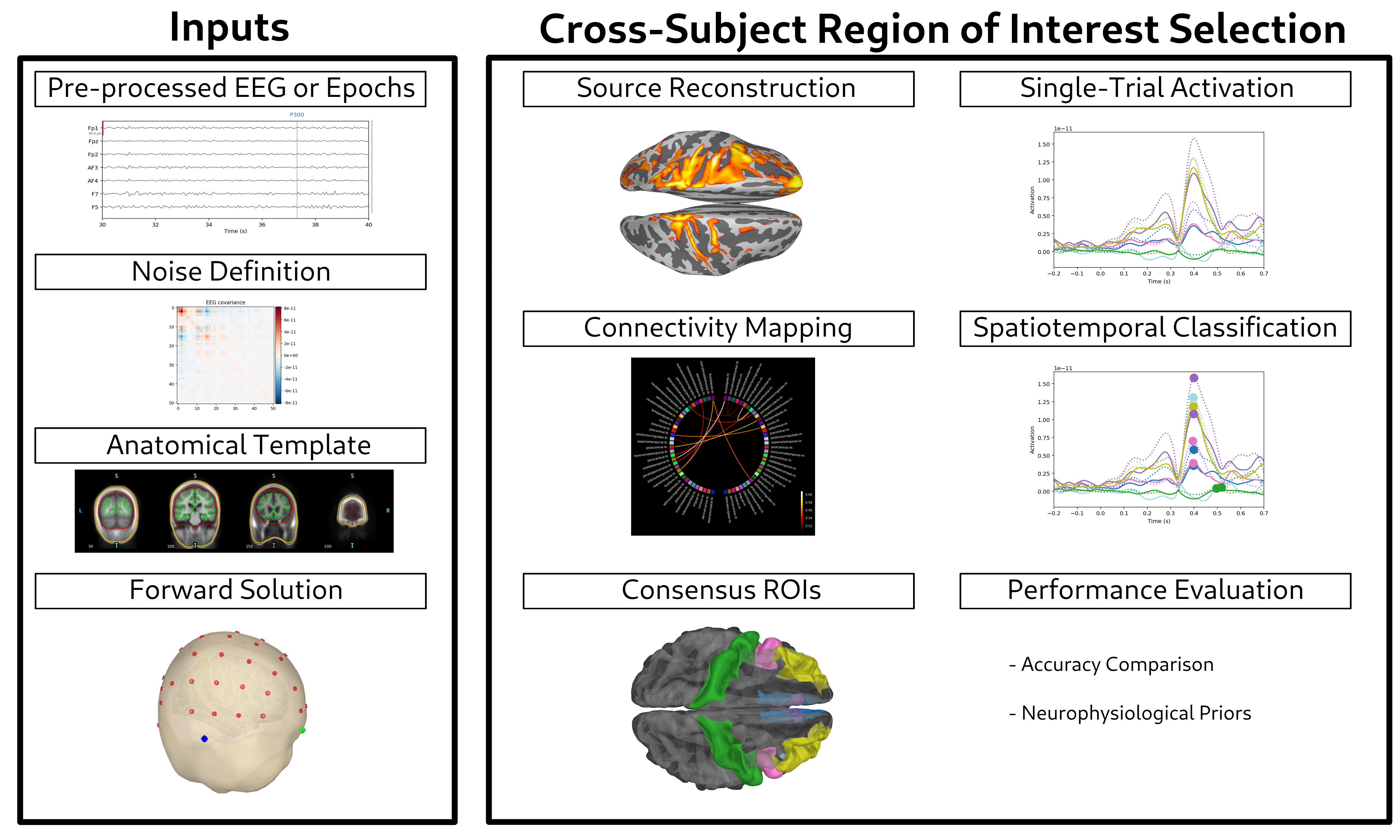
Figure 1: Consensus-Driven Hybrid Framework for Single-Trial P300 Detection Based on Cross-Subject Region of Interest (ROI) Selection.
Methodological Framework
Signal Preprocessing and Source Localization
The study utilizes the Eye-BCI multimodal dataset, comprising 62-channel EEG recordings from 31 subjects across 2,520 P300 trials. The preprocessing pipeline includes artifact correction (ABCD algorithm), cubic spline drift removal, Laplacian spatial filtering, and zero-phase Butterworth bandpass filtering to isolate canonical frequency bands. Epoch segmentation (−200 to +700 ms) preserves pre-stimulus baselines for normalization.
Source localization is performed using eLORETA, which provides voxel-wise reconstructions of cortical generators without a priori assumptions. The FreeSurfer fsaverage template and MNE-Python's BEM are employed for anatomical modeling, with a discretized source space of 10,242 dipoles per hemisphere. This configuration balances spatial resolution and computational efficiency, enabling reliable ROI identification.
Functional Connectivity and ROI Consensus
Functional connectivity is assessed via phase synchronization metrics—PLI, wPLI, and dPLI—across FreeSurfer's 68-region parcellation. Time-locked extraction during P3a (150–250 ms) and P3b (300–500 ms) windows isolates network interactions relevant to P300 generation. Stable ROIs are defined as regions most consistently observed among the top 20 connection pairs across sessions, ensuring cross-subject generalizability.
Single-trial classification is based on activation maxima within ROIs, with trials labeled P300-positive if their maxima fall within the neurophysiologically plausible window (+200 to +550 ms). Additional criteria include temporal agreement (maxima clustering) and a hybrid approach combining spatial and temporal validation.
Results
The hybrid ROI-driven framework demonstrates superior accuracy compared to whole-brain approaches across all frequency bands and connectivity metrics. Notably, PLI and dPLI achieve the highest accuracy in the 1–15 Hz band (81.57% and 79.90%, respectively), outperforming the state-of-the-art mean (73.31% ± 17.39%). wPLI, while less specific to P300 mechanisms, exhibits enhanced robustness in theta and beta bands due to its magnitude-weighted phase difference computation.
Neurophysiological Insights
Connectivity analysis reveals that phase-lagged delta–theta coupling binds frontal and insular sources for attentional capture (P3a), while alpha–theta coherence between temporal and parietal cortices sustains context-updating (P3b). dPLI uncovers a posterior executive hierarchy, with top-down modulation from the isthmus cingulate and bottom-up signaling from primary visual areas. wPLI identifies beta-band coherence in frontal and supramarginal regions, supporting sustained attention and multisensory integration.
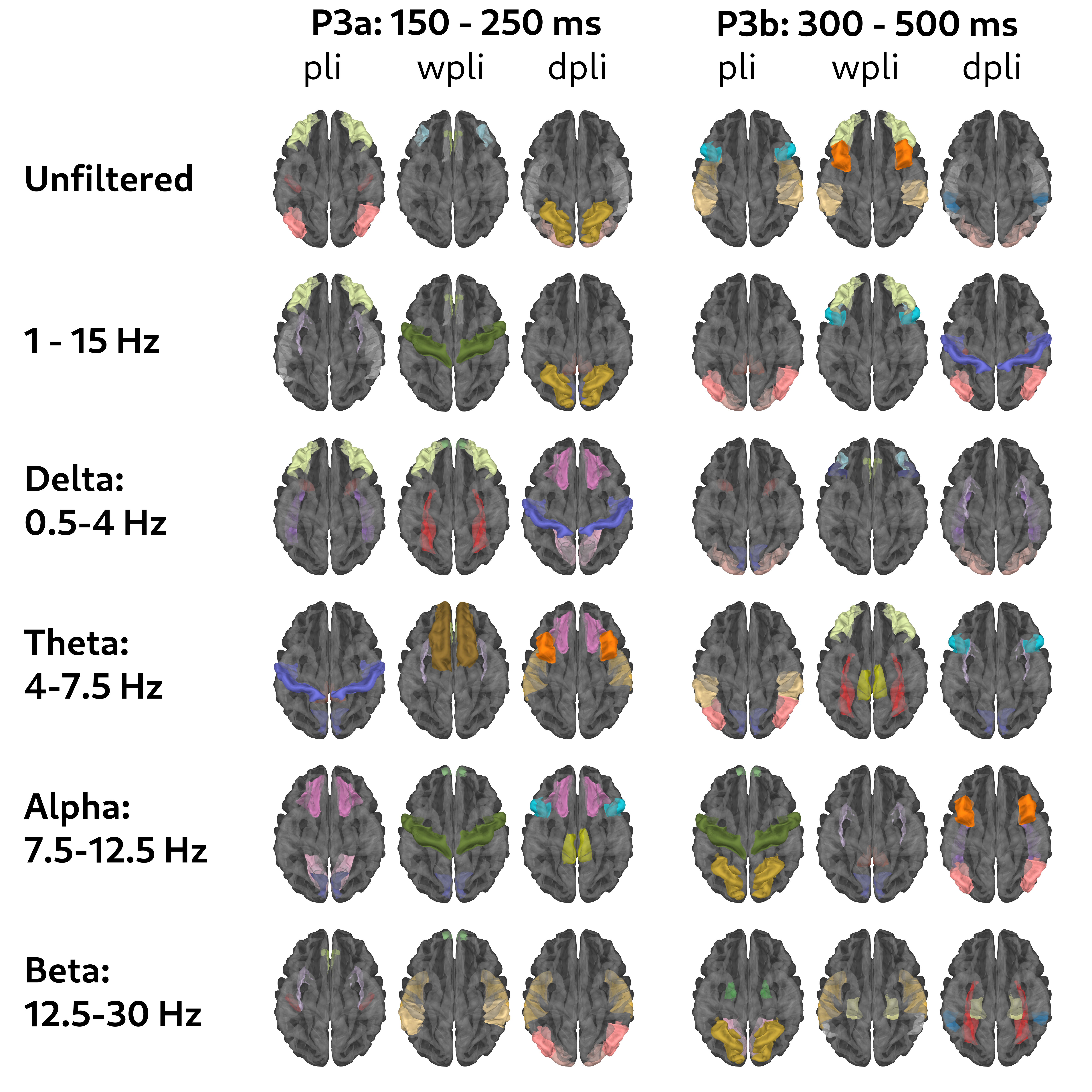
Figure 2: Spatiotemporal Dynamics of P300: Cross-Subject Functional Hubs via Phase Synchronization.
Discussion
Theoretical and Practical Implications
The framework's integration of eLORETA-based ROI selection and phase-resolved connectivity metrics provides mechanistic interpretability and robust classification performance. By isolating disease-relevant hubs (insula, parietal), the approach enhances the detection of neurodegenerative biomarkers, particularly for MCI and AD. The method's insensitivity to cultural and educational biases, coupled with compatibility for portable systems, positions it for global clinical deployment.
The study highlights the importance of spatial selectivity in EEG-based BCIs, demonstrating that indiscriminate whole-brain pooling degrades separability and introduces systematic error. The hybrid spatiotemporal criterion effectively addresses inter- and intra-subject variability, preserving millisecond-level temporal precision.
Limitations and Future Directions
While the framework achieves strong accuracy with standardized parameters, further refinement is possible through individualized anatomical modeling and adaptive parameter optimization. Future research should explore dynamic ROI cardinality and temporal windows based on subject-specific connectivity profiles, as well as multimodal validation against established biomarkers. Longitudinal studies in clinical populations will be critical for translating these findings into precision diagnostic tools.
Conclusion
The consensus-driven hybrid framework for single-trial P300 detection demonstrates that data-driven ROI selection, grounded in eLORETA source localization and cross-subject functional connectivity, significantly enhances classification accuracy in EEG-based BCIs. By targeting neurophysiologically relevant hubs and integrating spatiotemporal validation, the approach resolves the spatial-noise trade-off and advances the field toward clinically actionable neurophysiological monitoring. Future integration of individualized anatomical refinements and multimodal biomarkers will further extend its diagnostic potential.

