- The paper demonstrates that a distinct absence prediction error (aPE) signal emerges in the auditory thalamus and cortex, separate from traditional feature prediction errors.
- It employs a probabilistic omission paradigm with high-resolution fMRI to isolate BOLD responses that inversely scale with expected tone presence across auditory pathways.
- The findings prompt an extension of predictive coding models and offer potential clinical insights into perceptual dysfunctions such as auditory hallucinations.
Dedicated Prediction-Error Signaling for Auditory Absence in Thalamic Circuits
Theoretical Framework and Motivation
Prediction error signals are central to models of sensory processing under the predictive coding paradigm, which propose that perceptual inference relies on probabilistic representations of expected sensory features. Traditionally, predictive coding formulations have focused on updating beliefs about stimulus features (e.g., pitch, intensity), interpreting prediction errors as the discrepancy between expected and observed sensory values. However, these models do not directly address how the brain updates beliefs about a stimulus's categorical presence or absence—a critical capability for distinguishing sensory omission from feature change.
This work introduces and formalizes "absence prediction error" (aPE): a dedicated prediction error signal that reflects the brain's reduction in belief regarding the presence of an expected stimulus. The authors hypothesize the existence of specialized circuits for aPE and posit that its cortical and subcortical encoding differs from canonical feature prediction error (fPE) mechanisms.
Experimental Paradigm and Hypotheses
To disambiguate aPE from fPE and entrainment effects, the authors designed a probabilistic omission paradigm involving sequences of repeated pure tones. In each trial, an omission—a missing tone—could occur at positions 4, 5, or 6 within a sequence of up to eight repetitions, with omission probabilities carefully balanced and made explicit to participants. Participants were tasked with reporting the position of each omission, ensuring attentional engagement.
Expectations regarding omissions were controlled and quantified using abstract rules that manipulated beliefs about stimulus presence at each omission position. The paradigm thus enabled isolation of BOLD responses reflecting aPE, specifically those scaling with the ideal observer's expected probability of tone presence prior to omission (Figure 1).
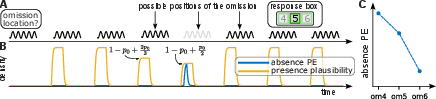
Figure 1: Experimental design enabling independent manipulation of omissions and statistical expectation, providing quantifiable predictors for aPE encoding.
Neural Responses to Omission and the Emergence of aPE
High-resolution 3T fMRI was used to measure BOLD activity in sequential auditory pathway components: the inferior colliculus (IC, midbrain), medial geniculate body (MGB, thalamus), and primary auditory cortex (PAC). Initial analyses confirmed robust adaptation to repeated standards across all ROIs, replicating established findings.
Critically, BOLD responses to unexpected omissions were significantly above baseline in bilateral PAC and MGB, but not in IC (Figure 2). This demonstrates for the first time the presence of omission-evoked responses in subcortical pathways.
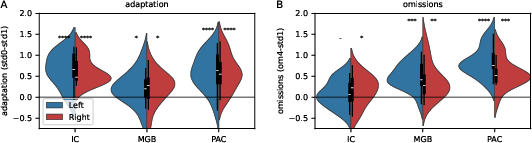
Figure 2: Violin plots depicting pronounced omission responses in thalamic and cortical ROIs, contrasting with weak or absent effects in midbrain nuclei.
Analysis of omission position responses revealed a negative scaling of BOLD signal with a priori expectation: higher plausibility of a tone (lower omission probability) led to larger evoked responses (Figure 3). This relationship is characteristic of aPE, supporting its novelty as a distinct neural computation.
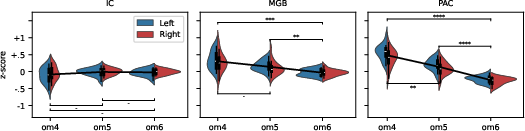
Figure 3: BOLD responses to omission positions inversely relate to expected presence, consistent with aPE encoding, and intensify along the ascending pathway.
To rigorously control for regional differences in BOLD sensitivity, responses were normalized using a localizer contrast and residual noise metrics (Figure 4, Figure 5). After normalization, adaptation effects were strongest in IC, yet omission and aPE responses were prominent in MGB and PAC, but entirely absent in IC.
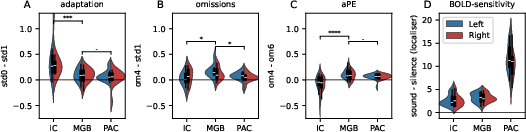
Figure 4: Pathway-wise comparison of normalized omission and aPE contrasts shows selective emergence of aPE in the thalamus and cortex.
Individual-Level Consistency and Group-Level Patterns
The group-level patterns were robustly reproduced at the individual participant level (Figure 6), with most subjects exhibiting significant aPE effects in MGB and PAC, but not in IC. This result suggests that aPE encoding is generalizable across subjects, providing strong evidence for its neural specificity and pathway localization.
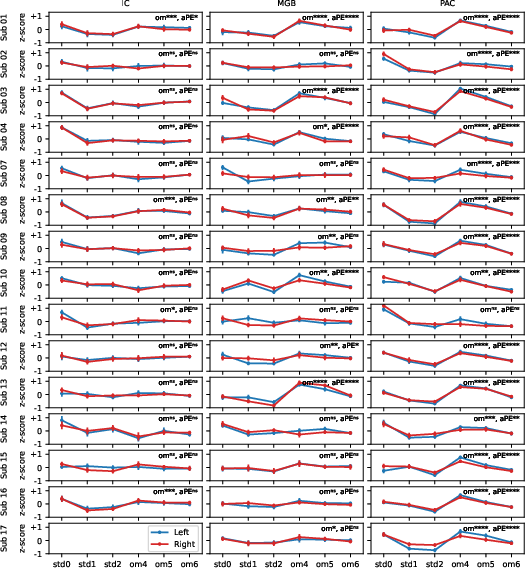
Figure 6: Single-subject BOLD contrasts confirm the systematic emergence of aPE in thalamic and cortical but not midbrain auditory regions.
Implications for Perceptual Inference and Theoretical Neuroscience
These findings necessitate a theoretical extension of predictive coding architectures to include distinct representations for stimulus presence/absence, beyond continuous-valued feature updates. The inability of Gaussian belief models (canonical in predictive coding) to represent stimulus absence is highlighted, motivating the need for more expressive probabilistic frameworks in perceptual inference models.
The dissociation between fPE and aPE along the ascending auditory pathway—fPE in both midbrain and thalamus, but aPE only in thalamus/cortex—suggests differential circuit-level computations for categorical perception. The authors speculate that negative prediction errors for presence (aPE) and positive prediction errors may operate in parallel, with possible anatomical specialization.
Clinical and Translational Perspectives
Disordered processing of presence and absence predictions is implicated in pathologies such as psychosis, where updating beliefs in the absence of expected stimuli (e.g., auditory hallucinations) is impaired. The identification of thalamic circuits encoding aPE provides a mechanistic substrate for studies targeting perceptual disturbances in clinical populations. Future work may leverage these markers for diagnostic or intervention strategies, and for examining the computational pathophysiology of psychiatric illness.
Future Directions in AI and Computational Neuroscience
The delineation of aPE prompts reconsideration of perception models in AI, particularly for architectures performing anomaly detection, out-of-distribution recognition, or learning under occlusion. Incorporating dedicated aPE-like signals could enhance agent robustness to absence detection, improving generalization and uncertainty estimation in sequential tasks. The observed circuit-level separation implies that distributed or hierarchical error signaling architectures may benefit from explicit absence-awareness modules.
Conclusion
This study establishes the existence of a dedicated absence prediction error signal that emerges in the human auditory thalamus and cortex but not in the midbrain. The aPE is distinct from classical feature prediction errors and is related quantitatively to the expectation of stimulus presence. These results advance our understanding of perceptual inference, underscore the need for expanded theoretical models, and provide new directions for investigating the neural basis of clinical perceptual dysfunction (2511.21605).




