- The paper demonstrates that incorporating higher-order interactions in multiplex networks yields a nonmonotonic vaccination coverage dependence on reinforcement, with optimal rates at intermediate values.
- Using a blend of homogeneous mean-field theory and well-mixed simulations, the authors show that social reinforcement modulates strategy switching, influencing epidemic size and vaccination clusters.
- Spatial patterns, including clustering of vaccinated agents into epidemic firebreaks, emerge from hyper-edge dynamics, highlighting practical implications for public health strategies.
Evolutionary Vaccination Dynamics under Higher-Order Reinforcement Pressure
This work analyzes evolutionary vaccination games embedded in multiplex networks, explicitly incorporating higher-order interactions (HOIs) in the behavioral updating process. The model consists of two distinct layers: a physical disease transmission network represented as a 2D square lattice, and a behavioral layer captured as a spatially embedded hyper-lattice featuring triadic (size-3) hyper-edges. Each agent participates in four overlapping hyper-edges (triangles), encoding group-level interactions that enable modeling beyond standard pairwise dynamics.
A season is defined by two phases: a strategy update (vaccination decision) among agents based on accumulated payoffs and peer influence, followed by an epidemic season governed by a classical SIR process that spreads only among non-vaccinated nodes. The decision process is a Fermi imitation rule, further modulated by a reinforcement parameter α when the focal agent and its observer share the same strategy, representing reinforcement pressure by peer consensus within the group.
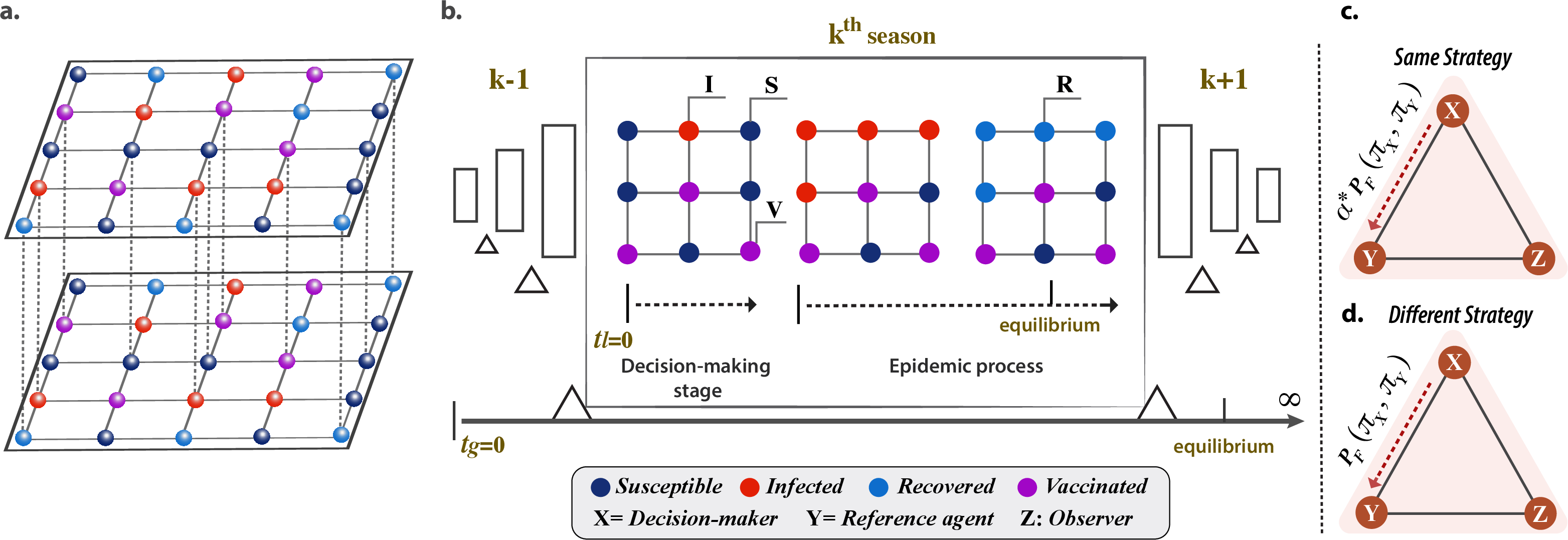
Figure 1: Schematic of the two-layer system: a behavioral hyper-lattice dictating strategy evolution (top) and a pairwise disease transmission network (bottom), with staged seasonal updates and reinforcement-moderated strategy adoption.
Strategies are binary (vaccinate or not), with payoffs parameterized by the relative cost of vaccination c (normalized against the infection cost). Non-vaccinators may free ride or incur losses if infected. Groups exhibit social reinforcement effects, as quantified by α, interpolating between unmodulated imitation (α=1) and maximal self-reinforcing inertia (α=0).
Homogeneous Mean-Field Theory and Well-Mixed Simulations
The authors construct a homogeneous mean-field theory (HMFT) that analytically tracks average vaccination coverage v(t) and disease progression by considering all possible triadic learner–reference–observer configurations and the resulting update probabilities under the Fermi rule scaled by reinforcement when appropriate. The SIR dynamics are standard, with initial vaccination coverage feeding as immune seeds into the epidemiological stage.
Simulation results in well-mixed populations show strong concordance with HMFT predictions: for fixed c, increasing α consistently reduces vaccination coverage and elevates the final epidemic size, as higher reinforcement pressure increases behavioral rigidity and slows adaptation.
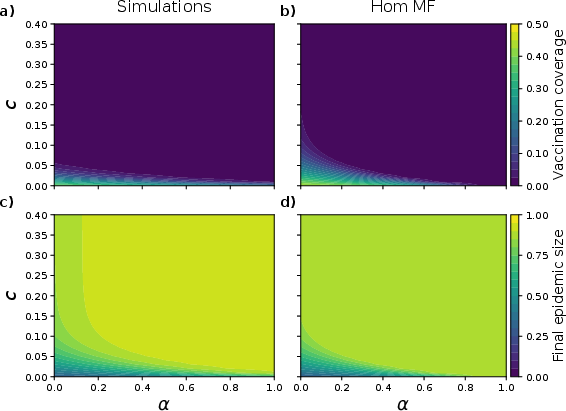
Figure 2: Macroscopic outcome comparison for well-mixed populations—simulation (left) vs. mean-field theory (right)—demonstrating near identity for vaccination coverage and epidemic size across (c,α).
Discrepancies between simulation and HMFT are negligible (<0.024% in FES), confirming the tractability and accuracy of the analytical model.
Role of Higher-Order Structure in Spatially Embedded Systems
Transitioning from well-mixed to structured multiplex systems, the authors unveil qualitatively distinct macroscopic phenomena. A key finding is the non-monotonic dependence of vaccination coverage on α. For all c, coverage is maximized at intermediate, low but nonzero α (approximately 0.05), while both extreme confirmation bias (α→0) and moderate-to-high reinforcement (α>0.1) suppress vaccination rates.
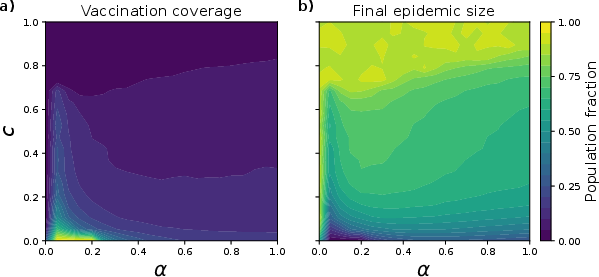
Figure 3: Vaccination coverage and epidemic size as heatmaps over (c,α) space for structured lattices, with a pronounced optimal α maximizing population-level immunity for fixed c.
These results reveal the critical interplay between higher-order neighborhood reinforcement and spatial topology: moderate reinforcement fosters consensus and rapid adoption, while excessive confirmation stabilizes fragmented or non-cooperative patches, permitting non-vaccinators to persist or re-emerge.
Analysis of spatiotemporal evolution elucidates the self-organization processes underpinning macroscopic outcomes. With α=0, vaccinated individuals aggregate into rectangular clusters that spatially shield susceptible populations and constrain disease percolation. Increase in α enhances the cohesion, speed, and prevalence of these clusters, accelerating the elimination of epidemic activity.
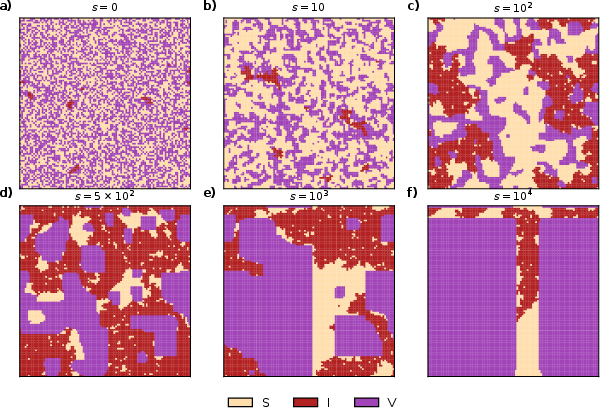
Figure 4: Spatiotemporal snapshots for α=0, c=0.05—the formation and growth of large, well-defined clusters of vaccinated individuals, acting as epidemic firebreaks.
At equilibrium, the shape, size, and prevalence of vaccinated clusters are strongly modulated by the vaccination cost; only low c yields extensive vaccine-dominated domains. Structural organization emerges from local consensus mechanisms inherent in hyper-edge-based reinforcement, even in the absence of explicit bias (i.e., α=0).
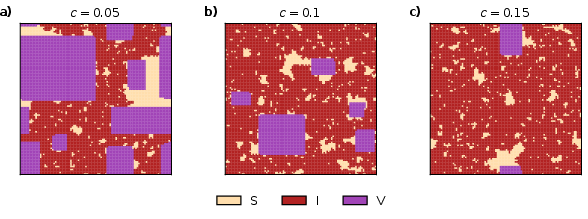
Figure 5: Equilibrium configuration dependence on c (with α=0): higher vaccination costs fragment vaccinated domains and reduce overall coverage.
Strategy Switching and Criticality of Reinforcement
A key innovation is the quantification of strategy transition probabilities across evolutionary time and parameter regimes. In baseline pairwise interaction scenarios, non-vaccinated clusters remain dominant except at very low cost. Higher-order (triadic) structure introduces oscillatory waves in transition probabilities, and the introduction of optimal reinforcement (α∼0.05) induces rapid and nearly complete convergence to universal vaccination.
However, as α is further increased (α=0.1), transition probabilities reveal a reversal: behavioral inertia locks in fragmented configurations, re-empowering non-vaccinators and decreasing coverage—a clear signature of nontrivial, resonance-like dependence on reinforcement strength.
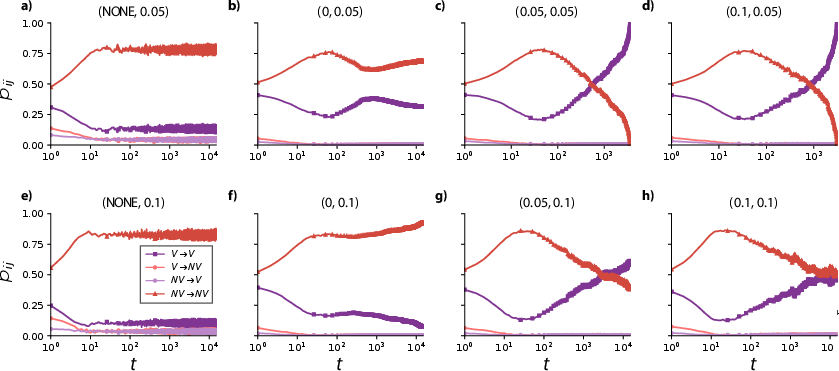
Figure 6: Evolution of behavioral transition probabilities, highlighting the emergence, amplification, or suppression of strategic switching and consensus as a function of (c,α).
Theoretical and Practical Implications
The study establishes critical connections between network topology, HOI-driven behavioral reinforcement, and epidemic control. Major implications include:
- HOIs alone, even without explicit reinforcement bias, induce nontrivial spatial patterning and can elevate vaccination rates vis-à-vis standard dyadic processes.
- Vaccination coverage is maximized at intermediate, low-but-nonzero social reinforcement (α), while both too much and too little confirmation suppress the uptake, reflecting a resonance effect.
- In structured populations, optimal reinforcement permits the achievement of herd immunity under cost regimes where classic pairwise models would predict endemicity.
These insights refine theoretical understanding of complex contagion, evolutionary games, and public health interventions by revealing how spatial structure and behavioral inertia nonlinearly interact. Practically, this suggests that public health messaging and policies should seek an intermediate level of consensus-building and group endorsement rather than maximal echo-chamber effects, to avoid locking populations into sub-optimal equilibria.
Future Directions
While the model abstractly formalizes triadic learning, future research should (i) empirically investigate the prevalence and measurable strength of higher-order social reinforcement mechanisms in real-world vaccination contexts, (ii) extend the analysis to heterogeneous, dynamic, or data-driven network topologies, and (iii) explore variable, time- or context-dependent reinforcement bias to reflect changing social attitudes or events.
Conclusion
This work presents a technical advance in the modeling of evolutionary vaccination games, embedding decision-making within higher-order, triadic, spatial structures and quantifying the emergent interplay of peer reinforcement and disease transmission. The main results robustly demonstrate that HOIs and their associated reinforcement effects can restructure both the macroscale behavioral landscape and the microscale evolution of cooperation—in this case, vaccination—yielding non-monotonic, sometimes counterintuitive, dependencies on the strength of social influence. This framework enhances the formal toolkit for epidemic control strategies design and complex systems analysis in public health (2601.12114).





