VERIDAH: Solving Enumeration Anomaly Aware Vertebra Labeling across Imaging Sequences
Abstract: The human spine commonly consists of seven cervical, twelve thoracic, and five lumbar vertebrae. However, enumeration anomalies may result in individuals having eleven or thirteen thoracic vertebrae and four or six lumbar vertebrae. Although the identification of enumeration anomalies has potential clinical implications for chronic back pain and operation planning, the thoracolumbar junction is often poorly assessed and rarely described in clinical reports. Additionally, even though multiple deep-learning-based vertebra labeling algorithms exist, there is a lack of methods to automatically label enumeration anomalies. Our work closes that gap by introducing "Vertebra Identification with Anomaly Handling" (VERIDAH), a novel vertebra labeling algorithm based on multiple classification heads combined with a weighted vertebra sequence prediction algorithm. We show that our approach surpasses existing models on T2w TSE sagittal (98.30% vs. 94.24% of subjects with all vertebrae correctly labeled, p < 0.001) and CT imaging (99.18% vs. 77.26% of subjects with all vertebrae correctly labeled, p < 0.001) and works in arbitrary field-of-view images. VERIDAH correctly labeled the presence 2 Möller et al. of thoracic enumeration anomalies in 87.80% and 96.30% of T2w and CT images, respectively, and lumbar enumeration anomalies in 94.48% and 97.22% for T2w and CT, respectively. Our code and models are available at: https://github.com/Hendrik-code/spineps.
Paper Prompts
Sign up for free to create and run prompts on this paper using GPT-5.
Top Community Prompts
Explain it Like I'm 14
Overview
This paper is about teaching a computer to correctly name the bones in your spine (the vertebrae) on medical scans like MRI and CT. Most people have 7 neck (cervical), 12 mid-back (thoracic), and 5 lower-back (lumbar) vertebrae. But some people naturally have one extra or one fewer in the thoracic or lumbar regions. These are called “enumeration anomalies.” The new method, called VERIDAH, can label vertebrae correctly even when these anomalies are present and even when the scan only shows part of the spine.
What questions did the researchers ask?
They set out to answer three simple questions:
- Can we build a computer system that labels every vertebra correctly, even when someone has an extra or missing thoracic or lumbar vertebra?
- Can it work well on both MRI and CT scans, including scans that don’t show the full spine?
- Is it more accurate than existing methods?
How did they do it?
The team combined two ideas: a smart classifier (a computer program that recognizes patterns) and a “sequence solver” (a rule-based checker that makes sure the final list of labels makes sense).
The data they used
- Thousands of 3D spine scans: about 6,300 whole-spine MRI scans and about 1,500 CT scans with many different views and qualities.
- Each vertebra’s location was known (from previous tools or expert-reviewed segmentations), so the system could “zoom in” on each bone.
Teaching the computer to look at each vertebra
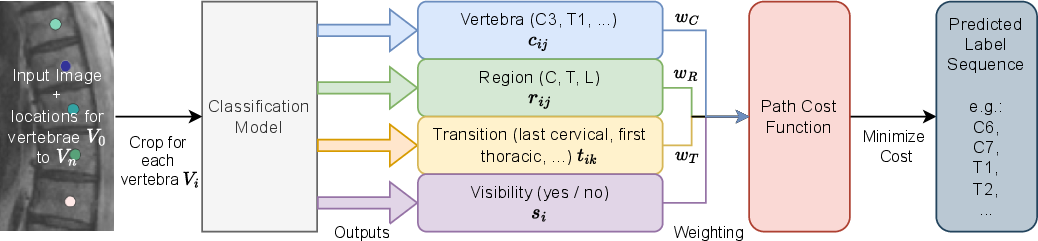
Think of the classifier like a group of four “mini-experts” that look at each vertebra’s close-up and vote on different things:
- Which exact vertebra is this? (like C3, T5, L1; where C is cervical, T is thoracic, L is lumbar)
- Which region is it in? (neck/cervical, mid-back/thoracic, lower-back/lumbar)
- Is it a special “transition” bone? (for example, the last thoracic or the first lumbar)
- How visible is it? (fully seen or partly cut off)
These four opinions help the system be more robust. For example, even if it’s unsure between T12 and L1, it might still be confident that the bone is in the thoracic region and is the “last thoracic,” which is a strong clue.
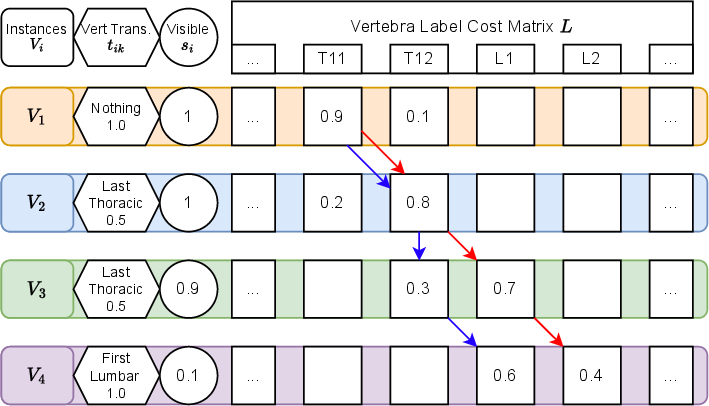
Choosing the best label sequence
After the mini-experts vote on each bone, another step checks that the final list of labels follows the rules of a spine. Think of it like solving a puzzle:
- Vertebrae must go in order from top to bottom (you can’t have T6 and then T5).
- The counts must make sense (normally 7 cervical, 12 thoracic, 5 lumbar—but the system also allows 11 or 13 thoracic and 4 or 6 lumbar to handle anomalies).
- In special cases, it allows patterns that mean “extra” bones (for example, two T12s in a row means there is actually a T13).
The system picks the sequence that best matches the mini-experts’ votes while obeying these rules. If a bone is barely visible, its vote counts less—like trusting a blurry photo less than a clear one.
What did they find?
VERIDAH was very accurate and beat other methods on both MRI and CT:
- On MRI: it labeled all vertebrae correctly in about 98% of people. It correctly detected thoracic anomalies in about 88% and lumbar anomalies in about 94%.
- On CT: it labeled all vertebrae correctly in about 99% of people. It correctly detected thoracic anomalies in about 96% and lumbar anomalies in about 97%.
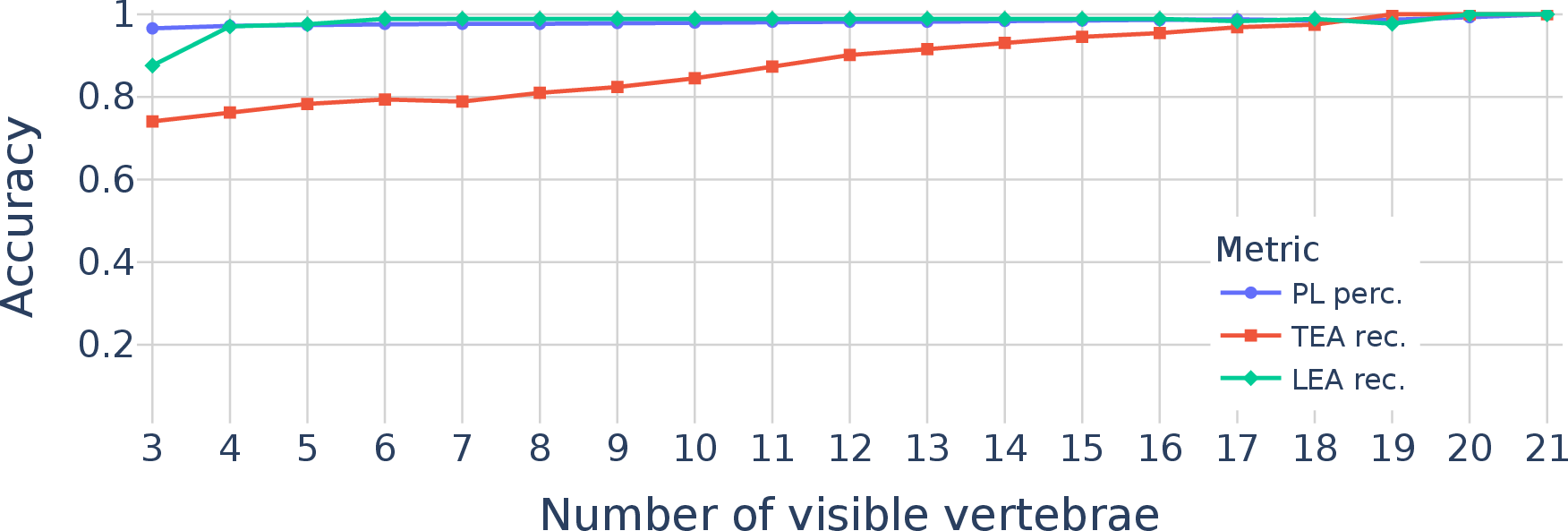
It also worked well even when the scan showed only part of the spine (a small field-of-view), which is common in real hospitals. Compared to other methods, it was especially better at finding people with extra or fewer vertebrae—something previous tools often missed.
Why is this important?
Correctly naming vertebrae matters in real life:
- It helps doctors plan surgeries safely and avoid operating on the wrong level.
- It supports consistent reports, which can reduce mistakes and save time.
- It can reveal anatomy that might relate to back pain or other issues.
By sharing their code and models, the authors make it easier for hospitals and researchers to use and build on their work. In short, VERIDAH makes spine labeling more reliable, especially when people’s anatomy doesn’t follow the “usual” numbers—something that really happens and really matters.
Knowledge Gaps
Knowledge gaps, limitations, and open questions
Below is a concise list of what remains missing, uncertain, or unexplored in the paper, framed to enable concrete follow-up work:
- End-to-end evaluation: VERIDAH requires precomputed vertebra centroids and is not assessed as part of a full pipeline (detection → labeling). Quantify performance under realistic detection errors (missing/mislocalized vertebrae, false positives/duplicates), including multiple gaps and noisy centroids.
- External validation for MRI: The T2w model is trained and evaluated on a single cohort (NAKO). Test generalization across vendors, magnet strengths, sequences (e.g., T1w, STIR), and multi-center datasets to establish robustness.
- CT validation without T1/sacrum: CT evaluation includes only scans with T1 visible (to allow manual TEA ground truth). Measure performance in partial CT FOVs where neither T1 nor sacrum is present—the common clinical scenario that motivated the method.
- Pediatric and complex anatomy: Assess performance in pediatric spines, severe deformities (scoliosis, kyphosis), postoperative instrumentation, fractures, block/hemivertebrae, and congenital variants that alter vertebral morphology beyond simple ±1 enumeration changes.
- Broader anomaly coverage: The constraints assume 11–13 thoracic and 4–6 lumbar vertebrae and only model T13/L6 via duplicated labels. Extend to rarer or combined anomalies (e.g., cervical ribs, sacral count variability, fused/missing vertebrae, multilevel transitional changes) and evaluate detection/labeling under these conditions.
- Transitional morphology (TLTV) characterization: The method labels enumeration anomalies but does not explicitly detect or classify transitional vertebrae types (e.g., facet orientation variants). Integrate rib/facet/articulation cues or dedicated segmentation to disambiguate TLTV and reduce failure modes at the thoracolumbar junction.
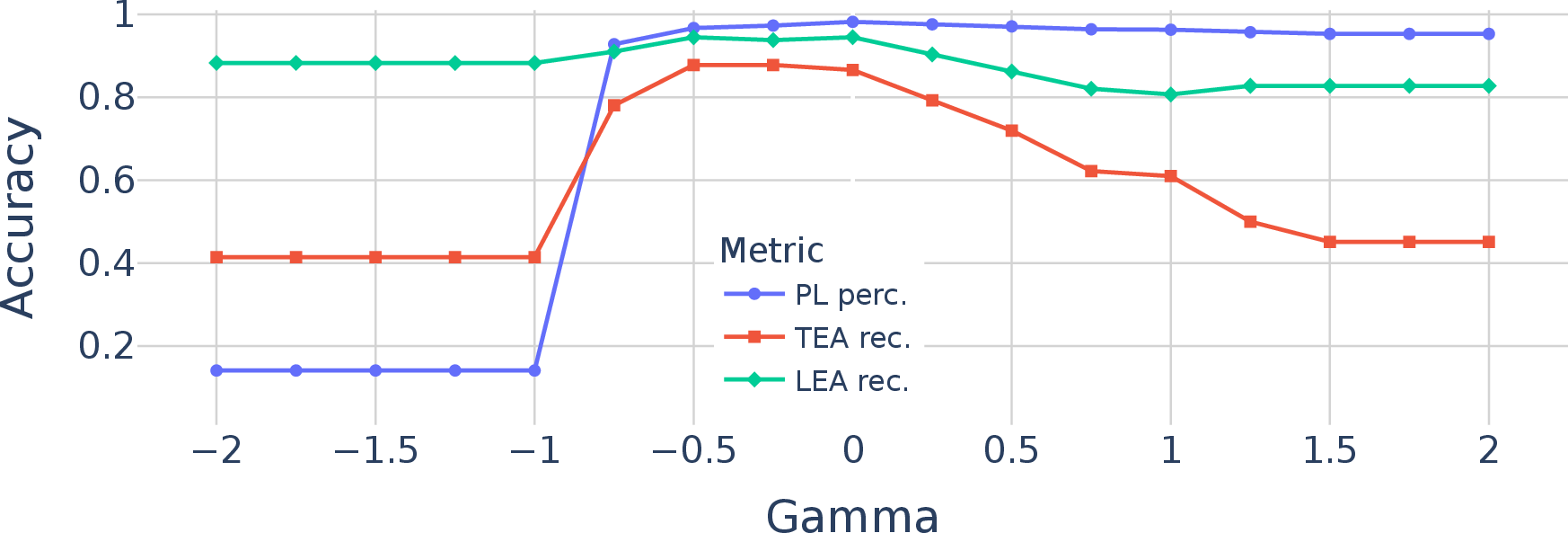
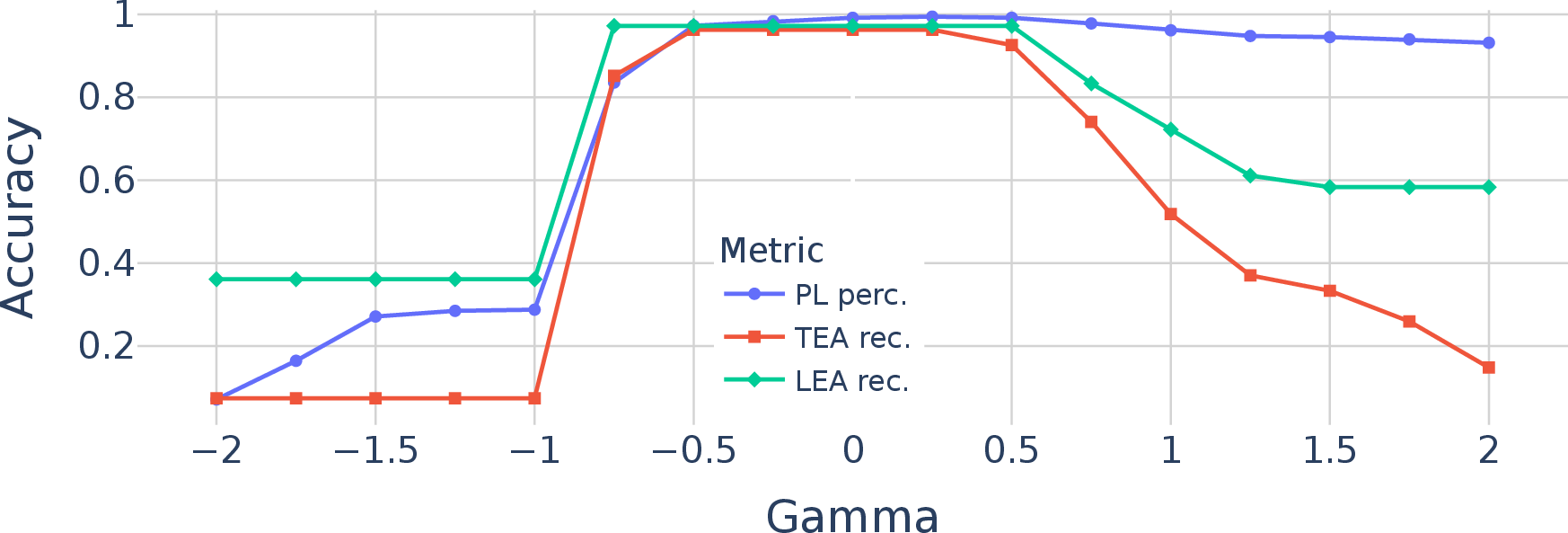
- TEA recall remains imperfect in MRI: With TEA recall at 87.8% (MRI), analyze failure cases and develop targeted improvements (e.g., rib detection modules, facet orientation estimation, TL junction-focused classifiers, enhanced constraints).
- Single-reader ground truth: All annotations were produced by one expert (3 years’ experience). Establish inter-rater reliability, multi-reader consensus, and adjudication to reduce label noise and quantify annotation uncertainty—especially for ambiguous TL junctions.
- Class imbalance handling: Anomaly cases were oversampled ×4. Systematically explore alternative strategies (class-balanced/focal losses, reweighting, synthetic augmentation) and quantify sensitivity of TEA/LEA recall to sampling ratios.
- Learned head fusion: Weights combining heads (w_C, w_R, w_T) are fixed via small calibration. Investigate learning these weights end-to-end, adaptive per-case weighting, or Bayesian fusion to better exploit multi-head signals under varying image quality and FOV.
- Uncertainty calibration: Softmax outputs are smoothed and combined but not calibrated. Implement per-vertebra and sequence-level uncertainty (e.g., temperature scaling, MC dropout) and use it to flag questionable labels for expert review.
- Constraint design: Current constraints enforce consecutive ordering and bounded counts; they force-fit anatomy into predefined ranges. Explore mechanisms to detect and communicate true outlier anatomies (instead of penalizing them) and to incorporate soft spatial priors (e.g., intervertebral distance, curvature) without overconstraining.
- Structured modeling comparison: Compare dynamic programming to alternative structured models (e.g., CRFs, HMMs, transformers with monotonic constraints) that can learn label transitions and anomaly patterns while providing uncertainty estimates.
- Visibility head robustness: The visibility-based weighting is heuristic and depends on Gaussian smoothing. Validate its calibration across varying SNR, motion artifacts, slice thickness, and different acquisition protocols; examine failure cases where low visibility leads to mislabeling.
- FOV-specific weaknesses: TEA recall degrades in small FOVs. Quantify minimal context requirements around the TL junction and design targeted modules (e.g., local rib detectors) to mitigate small-FOV performance drops.
- Runtime and deployment metrics: Training time is reported, but inference latency, memory footprint, and throughput are not. Provide deployment benchmarks and optimizations for clinical integration (e.g., batching, tiling strategies, GPU/CPU performance).
- Cross-modality/domain adaptation: Separate models are trained for CT and MRI. Evaluate domain adaptation, multi-modal training, and cross-modality transfer to reduce data requirements and improve generalization.
- Orientation and preprocessing variability: The pipeline assumes consistent reorientation and, for MRI, stitching of multi-slab acquisitions. Test robustness to orientation deviations, stitching artifacts, and vendor-specific pre-processing differences; provide generalized preprocessing guidelines.
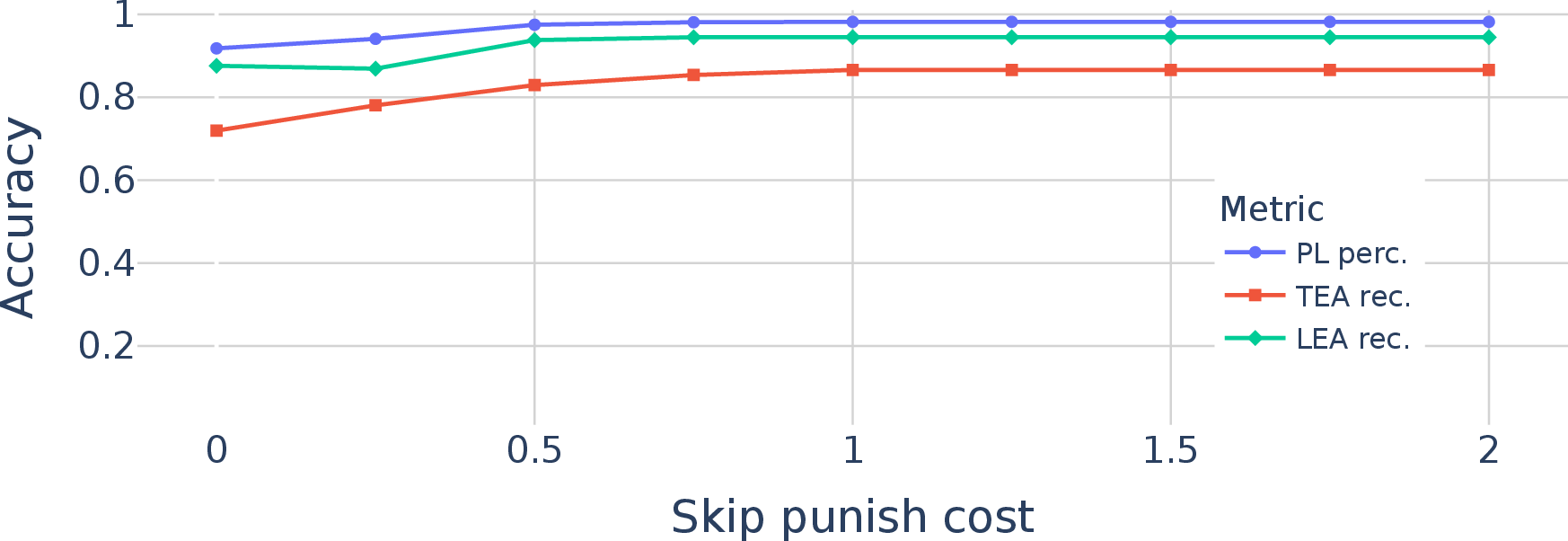
- Gap handling beyond single omissions: Ablations simulate one missing vertebra per subject. Extend experiments to multiple gaps, adjacent gaps, and false positives, reflecting real detection outputs; quantify sequence predictor performance under these conditions.
- Effect of contrast and protocol diversity (CT/MRI): The CT cohort includes both contrast-enhanced and non-contrast scans, but no stratified analysis. Measure performance across contrast use, reconstruction kernels, dose levels, and MRI scan parameters (e.g., echo spacing).
- Sacral labeling and pelvis context: The model stops at L6 and does not model sacral vertebrae (S1–S5) or pelvic anatomy. Explore extending labels into the sacrum and integrating pelvic landmarks to improve LEA decisions (e.g., sacralization vs. lumbarization).
- Failure taxonomy and corrective strategies: Provide a systematic catalog of failure modes (e.g., TLTV misclassification, label shifts, small-FOV confusion) with targeted remedial methods and prioritize them by clinical impact.
- Clinical utility and safety: Quantify downstream impact (e.g., reduction of wrong-site surgery risk, improved fracture reporting consistency) via prospective or retrospective clinical studies; define thresholds for human-in-the-loop review based on uncertainty.
- Reproducibility with public data: CT evaluations rely on an in-house dataset; MRI stitching and SPINEPS segmentation are prerequisites. Supply end-to-end recipes using public datasets (e.g., VerSe) and alternative open detection/segmentation models to facilitate replication and adoption.
Practical Applications
Immediate Applications
Below are applications that can be deployed now using the paper’s publicly released models and code, together with standard spine segmentation/detection tools.
- Healthcare (Radiology) — Anomaly-aware vertebral labeling in CT and T2-weighted sagittal MRI
- Use case: Auto-label vertebrae in routine spine imaging, including thoracolumbar enumeration anomalies (TEA: 11 or 13 thoracic; LEA: 4 or 6 lumbar), to reduce reporting errors and improve consistency in arbitrary field-of-view scans.
- Tools/workflow: VERIDAH model integrated with existing vertebra localization (e.g., SPINEPS or VerSe-like detectors), run as a PACS/RIS plugin or a DICOM post-processing service; export vertebral labels in DICOM SR or embedded overlays.
- Assumptions/dependencies: Requires accurate vertebra centroids from a detection/segmentation step; current performance validated for T2w TSE sagittal MRI (NAKO cohort) and diverse CTs; small FOVs may lower TEA recall; human QA recommended for edge cases and transitional morphology.
- Healthcare (Pre-operative planning and safety) — “Wrong-level” surgery prevention checks
- Use case: Pre-operative verification of target vertebral level against anomaly-aware labeling to minimize wrong-site/wrong-level spine surgery.
- Tools/workflow: Pre-op imaging processed through VERIDAH; auto-generated verification checklist attached to the case; discrepancy alerts if manual and automated labels differ.
- Assumptions/dependencies: Integration with surgical planning systems and institutional safety workflows; regulatory and clinical governance for human-in-the-loop confirmation.
- Healthcare (Teleradiology and QA) — Second-reader audit for label consistency
- Use case: Automatic cross-check of manual vertebral numbering in remote reads or high-volume centers; flagging of potential TEA/LEA for radiologist review.
- Tools/workflow: Batch processing of incoming studies, dashboard to track flagged anomalies and inter-reader agreement.
- Assumptions/dependencies: Reliable upstream centroids; institutional acceptance of AI-based QA; known lower performance in very limited FOVs.
- Academia (Data curation and cohort harmonization) — Rapid, reproducible labeling at scale
- Use case: Standardize vertebral labels across large cohorts for downstream research (e.g., fracture prediction, degenerative changes), including anomaly-aware correction.
- Tools/workflow: Pipeline combining segmentation (SPINEPS for MRI or similar CT models) with VERIDAH; store labels as structured metadata; harmonize multi-center datasets.
- Assumptions/dependencies: Availability of compute and storage; adherence to shared anomaly definitions; quality control for rare transitional cases.
- Academia (Epidemiology and association studies) — TEA/LEA prevalence and phenotyping
- Use case: Automated identification of enumeration anomalies to quantify prevalence and study associations with chronic back pain, degenerative findings, or surgical outcomes.
- Tools/workflow: Retrospective processing of cohorts (e.g., NAKO-like datasets); statistical pipelines for correlational analyses; anomaly stratification in models.
- Assumptions/dependencies: Cross-cohort generalization; adequate sample sizes for anomalies; standardized morphological criteria for transitional vertebrae.
- Software (Medical imaging platforms) — Microservice/API for anomaly-aware labeling
- Use case: Offer VERIDAH as a REST API for clinical viewers or research platforms (e.g., MONAI-based apps), returning ordered vertebral labels with anomaly flags.
- Tools/workflow: Containerize VERIDAH; expose endpoints; integrate with viewers (OHIF, 3D Slicer) and DICOM toolkits.
- Assumptions/dependencies: GPU resources for 3D DenseNet inference; upstream localization module; privacy/security compliance.
- Education (Radiology training) — Case libraries with verified anomaly labels
- Use case: Build annotated teaching collections highlighting enumeration anomalies and transitional vertebrae to improve trainee recognition and numbering accuracy.
- Tools/workflow: Batch labeling of institutional archives; curated case sets; interactive quizzes and feedback using automated ground truth.
- Assumptions/dependencies: Faculty oversight for edge cases; alignment with local educational objectives.
- Operations (Quality metrics) — Department-level labeling accuracy tracking
- Use case: Monitor and improve vertebral labeling accuracy across teams, quantify anomaly detection rates, and reduce discrepancy-driven addenda.
- Tools/workflow: QA dashboards; periodic reviews; feedback loops into training and protocols.
- Assumptions/dependencies: Governance for performance monitoring; willingness to adopt anomaly-aware standards.
Long-Term Applications
These applications require additional research, scaling, validation, productization, or regulatory processes before broad deployment.
- Healthcare (End-to-end automation) — Full pipeline from raw imaging to anomaly-aware labeling across modalities and scanners
- Use case: A turnkey system that detects vertebrae, labels them with anomaly awareness, and handles missing vertebrae or artifacts robustly across MRI sequences beyond T2w TSE (e.g., T1, STIR), CT, and possibly X-ray.
- Dependencies: Stronger cross-modality generalization, broader multi-vendor datasets, more robust handling of gaps and artifacts, clinical validation studies, regulatory clearance.
- Healthcare (Advanced surgical navigation and robotics) — Intraoperative level verification
- Use case: Real-time anomaly-aware vertebral labeling aligned with intraoperative imaging/navigation to verify target levels in robotic and instrumented procedures.
- Dependencies: Real-time inference; tight integration with navigation systems; robust registration; regulatory approvals; human-in-the-loop workflows.
- Healthcare (Comprehensive spine AI suites) — Downstream anomaly-aware models
- Use case: Build vertebra-aware AI for fracture detection, disc degeneration grading, stenosis assessment, and intervention planning that natively accounts for TEA/LEA.
- Dependencies: Multitask training pipelines; standardized outputs (labels, discs, regions); large-scale annotated datasets with anomalies; prospective studies demonstrating clinical impact.
- Academia and Policy (Standards and guidelines) — Mandating anomaly checks in spine reporting and pre-op workflows
- Use case: Incorporate enumeration anomaly checks into radiology reporting templates and surgical safety checklists; standardize numbering conventions across institutions.
- Dependencies: Consensus statements by professional societies; interoperability standards (DICOM SR, FHIR); policy adoption; training and auditing frameworks.
- Software (PACS-native products) — Commercial-grade anomaly-aware labeling modules
- Use case: Vendor-supported PACS plugins offering robust vertebral labeling with service-level guarantees and support.
- Dependencies: Productization, performance guarantees on diverse scanners, support contracts, cybersecurity compliance; multi-site validation.
- Public Health and Finance (Risk reduction and reporting) — Lower litigation risk from wrong-level surgeries
- Use case: Use anomaly-aware verification to reduce adverse events and related costs; institute-level risk dashboards; insurer incentives for adoption.
- Dependencies: Demonstrated reductions in wrong-level events; payer/provider alignment; legal and compliance frameworks.
- Cross-anatomy AI (Methodological generalization) — Multi-head classification plus constrained sequence prediction for other ordered anatomical structures
- Use case: Apply the VERIDAH paradigm to label ribs, dental arches, intervertebral discs, or vessel branching sequences with anomaly awareness.
- Dependencies: Domain-specific datasets and constraints; adaptation of visibility weighting and dynamic programming to new structures; validation per anatomy.
- Education and Public Engagement (Interactive simulators) — Anomaly-aware training simulators and patient education
- Use case: Simulation environments for trainees to practice numbering with anomalies; patient-friendly visualizations explaining their unique anatomy for shared decision-making.
- Dependencies: UX design, validated instructional content, localization to languages/cultures; clinical oversight.
- Research (Fine-grained anomaly taxonomies) — Beyond coarse enumeration to transitional morphology
- Use case: Develop classifiers that capture nuanced transitional features (facet orientation, riblets, Castellvi grading) and their clinical implications.
- Dependencies: High-quality expert labels, consensus morphological criteria, multi-sequence imaging, prospective outcome data.
Common Assumptions and Dependencies Across Applications
- Upstream vertebra localization is required; performance depends on the accuracy of centroids/segmentations.
- FOV should include informative context (thoracolumbar junction for TEA, lumbosacral for LEA); very limited FOVs reduce anomaly recall.
- Current validation: T2w TSE sagittal MRI (NAKO cohort) and diverse CT; generalization to other sequences/scanners requires testing.
- Human oversight is advised, especially for ambiguous transitional vertebrae.
- Compute/GPU resources and integration effort (PACS/RIS, DICOM/FHIR) are necessary for clinical deployment.
- Regulatory approvals are needed for high-stakes clinical use (pre-op verification, intraoperative guidance).
Glossary
- Ablation studies: Controlled experiments removing or altering components to assess their impact on performance. "In this section, we conduct ablation studies to analyze our proposed VERIDAH approach in more detail."
- Accessory ossification centre: An additional bone formation center near a vertebra that can mimic rib-like structures. "If there is only a unilateral or bilateral accessory ossification centre (comparable to Type 4 as defined in \cite{park2016thoracolumbar}), we determined this to be a lumbar vertebra."
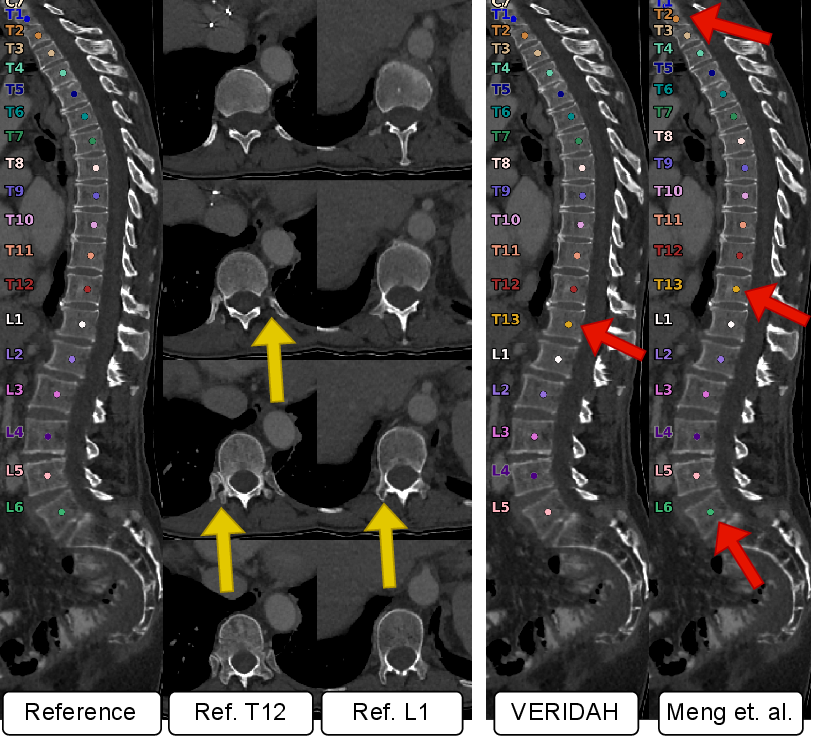
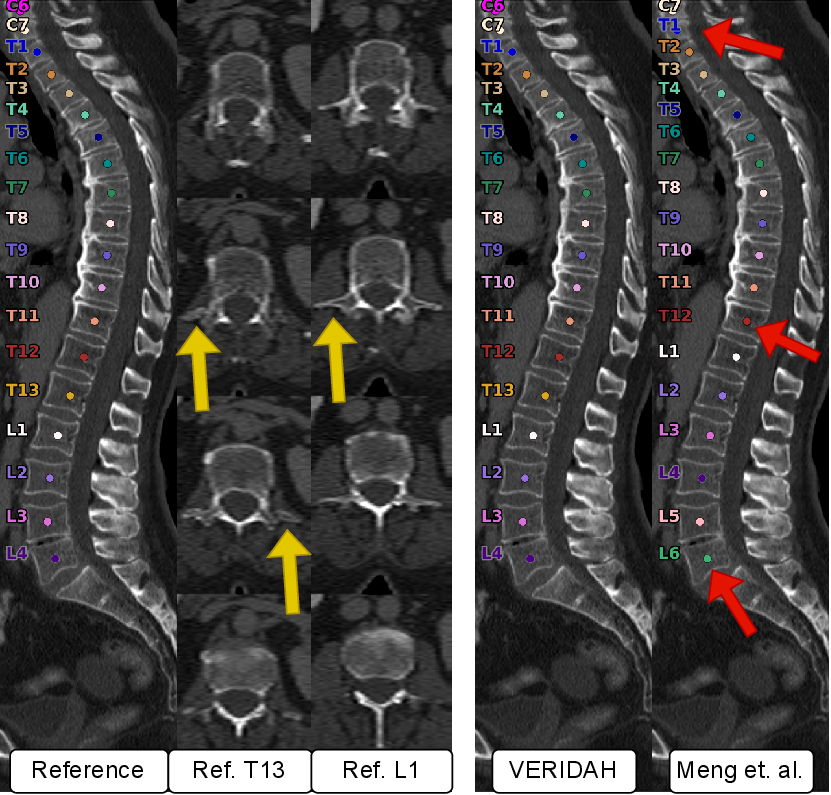
- Axial slices: Image sections taken in the transverse plane, perpendicular to the spine’s long axis. "The center shows the reference T12 and L1 in multiple axial slices (top to bottom)."
- Castellvi anomaly: A classification of lumbosacral transitional vertebrae based on morphologic types. "In the presence of a lumbosacral transitional Castellvi anomaly, we defined the vertebra with this anomaly as a sacralized lumbar vertebra regardless of the Castellvi type."
- Conditional random field: A probabilistic graphical model for structured prediction, often used to enforce label consistency. "Sekuboyina et al. \cite{sekuboyina2023pushing} developed a method for localization and identification of vertebrae in CT, using a conditional random field and user input to refine errors."
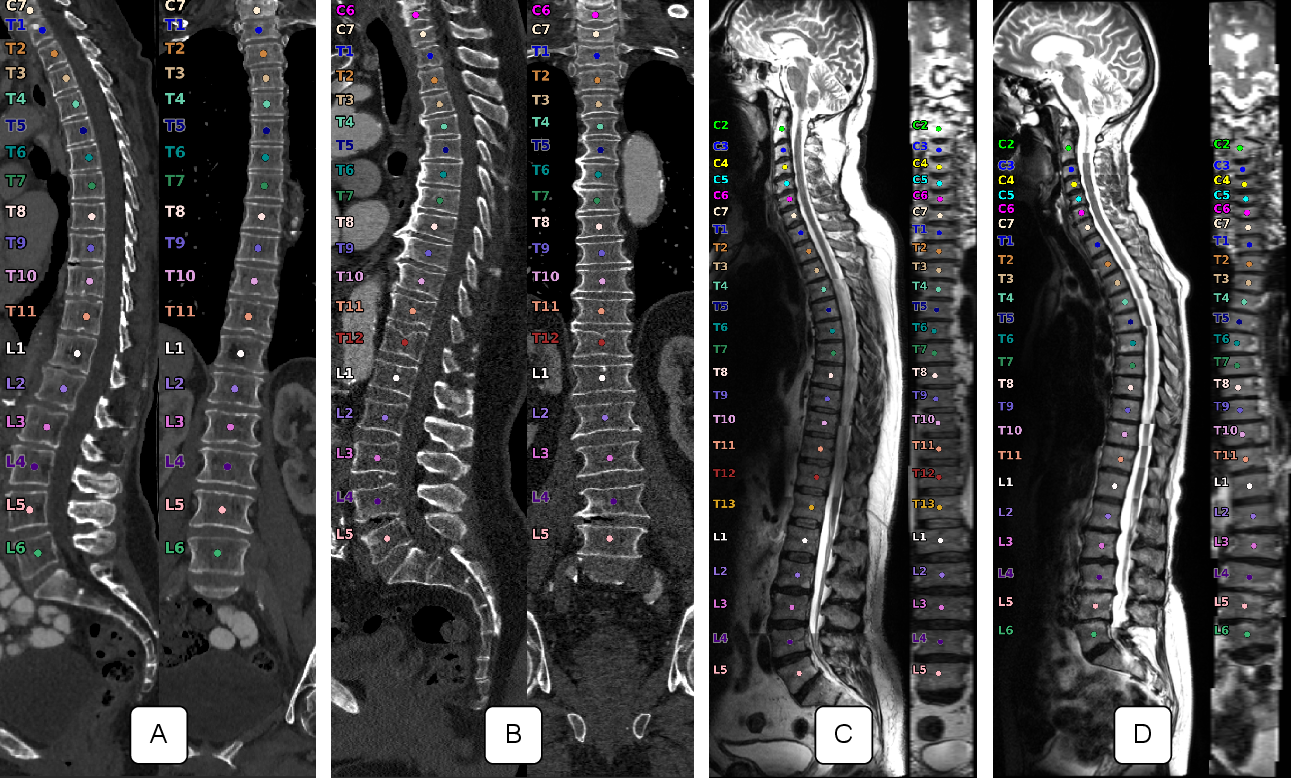
- Coronal projection: A view or projection of the image along the coronal (frontal) plane. "For each sample, it shows a sagittal and coronal projection along the spine as well as the corpus centroids with vertebra labels associated with each."
- Corpus centroid: The center point of the vertebral body (corpus), used for localization. "For each sample, it shows a sagittal and coronal projection along the spine as well as the corpus centroids with vertebra labels associated with each."
- Cranial-to-caudal: Anatomical direction from head (cranial) toward tail/feet (caudal). "A naive approach to identifying vertebrae is to count them in a cranial-to-caudal or caudal-to-cranial order."
- Dynamic programming: An optimization technique solving complex problems by combining solutions to subproblems. "In order to do so, we utilize dynamic programming and recursively run over all possible path options and compute the cost."
- Enumeration anomalies: Variations in the number of vertebrae in a spinal region (e.g., 11 or 13 thoracic). "However, there can be enumeration anomalies, causing a deviation of the number of thoracic and/or lumbar vertebrae by one"
- Facet orientation: The alignment of facet joints, used to distinguish thoracic vs. lumbar morphology. "In ambiguous cases, we judged by the morphology (e.g. facet orientation) whether a vertebra is thoracic."
- Field of view (FOV): The spatial extent covered by an imaging scan. "This requires a known reference point within the field of view (FOV)"
- Hyperintense spots: Areas appearing brighter than surrounding tissue on certain MRI sequences. "We did not observe obvious reasons for these failures, such as hyperintense spots or pathologies."
- Intervertebral discs: Fibrocartilaginous cushions between adjacent vertebral bodies. "Finally, Warszawer et al. \cite{yehudawarszawer202514920281} locate specific intervertebral discs during the task of segmentation in order to identify the sequence of vertebrae visible in the image by counting from those discs in both directions."
- Isotropic resolution: Voxel dimensions equal in all three spatial directions. "We resampled the CT images consistently to an isotropic (1.0, 1.0, 1.0) mm resolution."
- Lumbar enumeration anomaly (LEA): A deviation in the number of lumbar vertebrae (e.g., 4 or 6). "These thoracic enumeration anomalies (TEA) and lumbar enumeration anomalies (LEA) are often accompanied by transitional anomalies"
- Lumbosacral transition: The anatomical junction between lumbar spine and sacrum. "For the lumbosacral transition, we adapted the analysis of Konin et al. \cite{konin2010lumbosacral}."
- Rib-bearing (thoracic): Characteristic of thoracic vertebrae that articulate with ribs. "The human spine typically consists of seven cervical, twelve rib-bearing thoracic, and five lumbar vertebrae \cite{thawait2012spine}."
- Sacralized lumbar vertebra: A lumbar vertebra partially or fully fused/assimilated to the sacrum. "In the presence of a lumbosacral transitional Castellvi anomaly, we defined the vertebra with this anomaly as a sacralized lumbar vertebra regardless of the Castellvi type."
- Sacrum: A triangular bone at the base of the spine, below lumbar vertebrae. "This requires a known reference point within the field of view (FOV), such as the first thoracic vertebra (identified by the presence of a rib) or the sacrum (to identify the last lumbar vertebra)."
- Sagittal (plane): The anatomical plane dividing the body into left and right portions. "The T2w sagittal images of the NAKO always show the whole spine split into three segments and thus contain up to 26 fully visible vertebrae"
- Thoracic enumeration anomaly (TEA): A deviation in the number of thoracic vertebrae (e.g., 11 or 13). "These thoracic enumeration anomalies (TEA) and lumbar enumeration anomalies (LEA) are often accompanied by transitional anomalies"
- Thoracolumbar junction: The boundary region where the thoracic spine transitions to the lumbar spine. "Although the identification of enumeration anomalies has potential clinical implications for chronic back pain and operation planning, the thoracolumbar junction is often poorly assessed and rarely described in clinical reports."
- Thoracolumbar transitional vertebrae (TLTV): Vertebrae at the thoracolumbar junction with mixed thoracic and lumbar features. "These thoracic enumeration anomalies (TEA) and lumbar enumeration anomalies (LEA) are often accompanied by transitional anomalies, such as thoracolumbar transitional vertebrae (TLTV)."
- Transitional anomalies: Abnormal vertebrae exhibiting mixed characteristics of adjacent spinal regions. "These thoracic enumeration anomalies (TEA) and lumbar enumeration anomalies (LEA) are often accompanied by transitional anomalies, such as thoracolumbar transitional vertebrae (TLTV)."
- T2-weighted (T2w): An MRI contrast weighting emphasizing differences in T2 relaxation times. "For the T2w sagittal MRI data, we utilized a random subset of 6291 subjects of the German National Cohort (NAKO)"
- Turbo spin echo (TSE): An MRI pulse sequence acquiring multiple echoes per excitation to accelerate imaging. "We show that our approach surpasses existing models on T2w TSE sagittal (98.30\% vs. 94.24\% of subjects with all vertebrae correctly labeled, )"
- Wilcoxon signed-rank test: A nonparametric statistical test for paired samples. "We employ the Wilcoxon signed-rank test for statistical significance, with a p-value indicating significance."
- Wrong-site surgery: Surgical intervention performed at an unintended anatomical site. "This can result in wrong-site surgery, where the intended vertebra is missed and adjacent vertebrae are operated on by mistake \cite{tatara2021changes}."
Collections
Sign up for free to add this paper to one or more collections.