Second law of thermodynamics in closed quantum many-body systems
Abstract: The second law of thermodynamics for adiabatic operations -- constraints on state transitions in closed systems under external control -- is one of the fundamental principles of thermodynamics. On the other hand, it is recently established that even pure quantum states can represent thermal equilibrium. However, pure quantum states do not satisfy the second law in that they are not passive, i.e., work can be extracted from them if arbitrary unitary operations are allowed. It therefore remains unresolved how quantum mechanics can be reconciled with thermodynamics. Here, based on our key quantum-mechanical notions of thermal equilibrium and adiabatic operations, we address the emergence of the second law for adiabatic operations in the thermodynamics limit. We first introduce infinite-observable macroscopic thermal equilibrium (iMATE); a quantum state, including pure states, is in iMATE if the expectation values of all additive observables agree with their equilibrium values. We also introduce a macroscopic operation as unitary evolution generated by a time-dependent additive Hamiltonian, which is regarded as corresponding to adiabatic operations. Employing these concepts, we show that no extensive work can be extracted from any quantum state in iMATE through any macroscopic operations. Furthermore, we introduce a quantum-mechanical form of entropy density such that it agrees with thermodynamic entropy density for any quantum state in iMATE. We then prove that for any initial state in iMATE, this entropy density cannot be decreased by any macroscopic operations, followed by a time-independent relaxation process. Our theory thus proves two different forms of the second law, by adopting macroscopically reasonable classes of observables, equilibrium states, and operations. We also discuss the time scales of macroscopic operations in these results.
Paper Prompts
Sign up for free to create and run prompts on this paper using GPT-5.
Top Community Prompts
Explain it Like I'm 14
What is this paper about?
This paper asks a big question: How does the second law of thermodynamics—rules like “you can’t get free energy” and “entropy doesn’t go down”—show up in large, closed quantum systems? Even when the system is in a pure quantum state (which normally doesn’t have much entropy), can it still behave like a warm, balanced system that follows thermodynamic rules?
The authors build a bridge between quantum mechanics and thermodynamics by carefully defining what “thermal equilibrium” and “allowed operations” mean at the macroscopic (big-picture) level. Then they prove two classic second-law statements for closed quantum many-body systems.
The big questions they ask
To make thermodynamics work inside quantum mechanics, the paper focuses on five simple-sounding questions:
- How should we define a quantum state that truly behaves like thermal equilibrium at the macroscopic level?
- What kinds of operations (changes over time) count as realistic “thermodynamic operations” in closed systems?
- Can we prevent “free energy extraction” (violating passivity) from pure quantum states that look thermal?
- Is there a way to define an entropy for any quantum state that matches thermodynamic entropy when the state looks thermal?
- Does this entropy obey the second law—does it avoid decreasing—under realistic macroscopic operations?
How they study the problem
The authors work with large spin systems on a lattice (imagine a giant grid where each site is a tiny quantum bit). They focus on macroscopic, physically meaningful things and avoid operations that are unrealistic for very large systems.
Here are the key ideas explained in everyday language:
Additive observables: big-picture averages
An “additive observable” is something you can add up over space—like the total magnetization (sum of small local magnetizations across the whole system). These correspond to the quantities thermodynamics cares about: energy density, particle number density, magnetization density, and so on. If you divide by the system size, you get a spatial average.
Macroscopic equivalence: same big-picture look
Two quantum states are called “macroscopically equivalent” if, for every additive observable you can build from local measurements, the two states have the same average in the large-size limit. Even if the states look different microscopically, they look the same when you only care about big, averaged quantities.
iMATE: infinite-observable macroscopic thermal equilibrium
A state is in iMATE if, for all additive observables (not just a few), its averages match those of a Gibbs (thermal) state at some temperature. Think of iMATE as an upgraded, stricter version of “thermal equilibrium” that checks agreement for infinitely many macroscopic quantities, not just a small list.
Macroscopic operations: realistic controls
A “macroscopic operation” is a time-dependent change driven by turning a small number of global knobs (like a uniform field or a few large-scale controls). The Hamiltonian (the energy rule that drives time evolution) changes in time, but only by adding or removing terms that couple to additive observables. Crucially, the total operation time is fixed and does not grow with system size—this keeps the operation physically reasonable.
There’s also a speed limit built into quantum mechanics (related to the Lieb–Robinson bound): local changes spread through the system at a finite speed. If you try to do wild, global-unitary tricks that need unrealistically long times, they won’t count as “macroscopic operations.”
Entropy from local snapshots
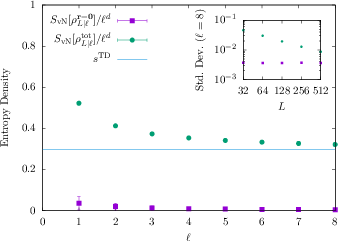
To define a meaningful entropy for any state, the authors take many small “local snapshots” of the state (reduced density matrices on small cubes), average them across space, and compute the von Neumann entropy of this spatially averaged local state. When the local cube size grows (but stays much smaller than the whole system), this “macroscopic entropy density” turns into the thermodynamic entropy density—if the state is iMATE.
Operations followed by relaxation
They study a process where you:
- Start in iMATE for the initial Hamiltonian.
- Apply a macroscopic operation for a fixed time.
- Stop changing the Hamiltonian and let the system evolve with a time-independent Hamiltonian, allowing it to relax. They then analyze the long-time average of the final state (the standard way to talk about equilibration in isolated quantum systems).
What did they find?
Here are the main results, explained simply:
- Macroscopic equivalence survives macroscopic operations
- If two states look the same at the big-picture level (same averages for all additive observables), and you apply any macroscopic operation for a fixed time, they still look the same afterwards. In short: reasonable, global controls don’t change the macroscopic identity of the state within normal time scales.
- Planck’s principle (macroscopic passivity) holds for iMATE
- You cannot extract an extensive amount of work (i.e., lower the energy density) from any iMATE state using allowed macroscopic operations that finish in a normal time. This is a version of “no free energy” for closed quantum systems that look thermal at the macroscopic level—even if the state is a pure state.
- At infinite temperature (β = 0), the energy density cannot change under these operations.
- If you allow operations that take longer and longer as the system grows, this protection can break—so the normal-time condition is essential.
- A quantum macroscopic entropy that matches thermodynamics
- The entropy density you get by averaging those local snapshots across space exactly equals the thermodynamic entropy density for any iMATE state. This gives a practical, quantum-friendly recipe to compute thermodynamic entropy from measurements of additive observables, even for pure states that look thermal.
- The law of increasing entropy under macroscopic operations plus relaxation
- If you start in iMATE, do any allowed macroscopic operation (within normal time), and then let the system relax under a fixed Hamiltonian, the macroscopic entropy density (from local snapshots) does not go down.
- If the final state also looks thermal (iMATE) for the final Hamiltonian, the result becomes the usual thermodynamic statement: the thermodynamic entropy does not decrease.
- Again, if you allow operations that take too long as the system grows, you can find counterexamples—so keeping the operation time independent of system size is optimal and necessary.
Why does this matter?
- It reconciles a puzzle: Pure quantum states can look thermal (by many tests) but are usually “non-passive” if you allow any unitary. This paper shows that, under realistic macroscopic operations and time constraints, the second law still holds—no extensive work can be extracted and entropy doesn’t decrease.
- It gives a clear, testable definition of thermal equilibrium for quantum states (iMATE) that matches thermodynamics not just for a few quantities, but for all additive observables.
- It provides a physically grounded notion of which operations are “thermodynamic” at the quantum level—macroscopic controls with finite, size-independent duration—rather than any mathematically possible unitary.
- It offers a practical entropy formula for quantum states that matches thermodynamics when the state is macroscopically thermal, including pure states.
- It strengthens the link between quantum mechanics and thermodynamics, helping us understand how irreversible behavior and entropy growth emerge from unitary quantum dynamics when we focus on macroscopic quantities and realistic operations.
In short, the paper shows that the second law of thermodynamics naturally emerges in closed quantum many-body systems—provided we look at the right macroscopic observables, use the right definition of equilibrium (iMATE), and restrict ourselves to realistic, normal-time operations.
Knowledge Gaps
Knowledge gaps, limitations, and open questions
Below is a concise list of unresolved issues and limitations that emerge from the paper’s definitions, assumptions, and results. Each item is framed to enable concrete follow-up research.
- Scope of interactions
- Extend results beyond short-range interactions with Lieb–Robinson bounds to systems with long-range couplings (e.g., power-law decays). Identify α thresholds and modified light-cone bounds under which macroscopic equivalence preservation and passivity still hold.
- Local Hilbert-space dimension
- Generalize from finite-dimensional spin systems to bosonic/continuous-variable models with unbounded local Hilbert spaces. Determine necessary conditions (e.g., energy cutoffs) for the entropy formula and passivity to remain valid.
- Integrability and additional conserved quantities
- Incorporate integrable systems and generalized Gibbs ensembles (GGE). Define an iMATE analogue consistent with multiple Lagrange multipliers and assess whether macroscopic passivity and the entropy law extend to GGE-based equilibria.
- Negative temperature states
- Clarify the status of macroscopic passivity for iMATE at negative inverse temperatures (). Determine whether a reversed inequality or a modified second-law statement holds, and under what operational constraints.
- Phase transitions and non-unique thermodynamic limits
- Analyze systems at first-order phase coexistence or with spontaneous symmetry breaking, where thermodynamic limits may be non-unique. Specify conditions under which iMATE and the entropy formula remain well-defined and second-law statements survive.
- Coarse-graining parameter K and spatial resolution
- The framework fixes the number of primitive macroscopic subregions independent of . Establish results that are uniform in , and explore the limit (finer macroscopic resolution) after . Identify where proofs break or require new bounds.
- Dependence on the order of limits in the entropy formula
- The entropy density uses an iterated limit . Investigate whether the order can be interchanged, conditions for uniform convergence, and the sensitivity of the result to different sequences of macroscopic subsystems.
- Finite-size and finite-time quantitative bounds
- Provide explicit finite-size error bounds for macroscopic equivalence preservation and passivity (e.g., dependence on , , Lieb–Robinson velocity). Quantify subextensive work that might still be extractable (e.g., ) and the dependence on protocol time.
- Operational class and physical realism
- Justify that thermodynamic operations are well-approximated by “macroscopic operations” (few additive controls, ), and characterize which standard adiabatic processes (compression, field sweeps, interaction changes) fit this class. Develop a systematic taxonomy of admissible thermodynamic operations that avoids the known counterexamples.
- Quasi-static and slow protocols
- Reconcile the prohibition of operation times scaling with (due to counterexamples) with quasi-static, reversible processes that are conventionally infinitely slow. Identify additional physical constraints (e.g., restricted bandwidth, locality of work source) that allow longer times without violating the second law.
- Spatially nonuniform controls at macroscopic scales
- Generalize operations to multiple macroscopic blocks with independently controlled additive generators (scaling with rather than a fixed ), and establish the largest admissible control class that still preserves macroscopic passivity.
- Relaxation dynamics vs long-time averages
- The final states are defined by long-time averages under time-independent . Derive sufficient conditions under which the actual dynamics approaches these time averages for relevant observables (dephasing, absence of revivals), and quantify relaxation timescales.
- Thermalization assumptions
- Convert the “mild assumption” about the existence of a unique thermodynamic limit into verifiable criteria. Provide ETH-based or alternative sufficient conditions that guarantee the final state represents iMATE, enabling a fully microscopic derivation of entropy increase in thermodynamic terms.
- Nonequilibrium meaning of the entropy for generic states
- Beyond iMATE, clarify the operational/physical meaning of the proposed entropy density for general nonequilibrium states (e.g., additivity, concavity, comparison with diagonal/coarse-grained entropies), and characterize its behavior under arbitrary (not necessarily macroscopic) unitaries.
- Robustness to weak long-range tails and experimental noise
- Assess how small deviations from strict additivity (e.g., weak long-range interactions) and realistic control noise affect macroscopic passivity and entropy increase. Provide stability theorems with explicit perturbative bounds.
- Inclusion of additional conserved charges and ensembles
- Extend the framework to grand-canonical settings (particle number, magnetization) with multiple control parameters and demonstrate second-law analogues involving corresponding thermodynamic potentials.
- Boundary conditions and geometry
- Prove that results are insensitive to boundary conditions and hold for open geometries with edges (quantify boundary contributions). Explore inhomogeneous lattices and non-cubic geometries.
- Fermionic systems and superselection
- Address fermionic anticommutation relations and superselection rules formally, ensuring that additive observables and macroscopic equivalence extend without hidden assumptions (e.g., Jordan–Wigner nonlocalities).
- Experimental measurability of the entropy
- Provide explicit measurement schemes for the entropy via additive observables: identify minimal sets of observables, reconstructive algorithms, sample complexity, and statistical error bounds for finite and finite .
- Nonunitary macroscopic operations
- Explore whether analogous second-law results hold for closed-system analogues of “adiabatic” but dissipative processes or weak coupling to work reservoirs without heat exchange. Connect with isothermal second-law results as a limit or extension.
- Microscopic models of work sources
- Derive the allowed macroscopic operations from explicit system–work source models, identifying conditions under which time-dependent additive Hamiltonians emerge and mapping resource-theoretic constraints to the paper’s control class.
- Strengthened passivity statements
- Investigate whether subextensive-work extraction is possible at intermediate timescales and search for strengthened versions of macroscopic passivity (e.g., bounds scaling as under additional constraints).
- Continuum and quantum field theories
- Translate the framework to continuum systems and quantum field theories where lattice structure and finite-dimensional local Hilbert spaces are absent. Establish analogues of additivity and Lieb–Robinson-type bounds (where available).
- Disorder and many-body localization
- Analyze how disorder and MBL (where thermalization fails) interact with macroscopic equivalence and the entropy law. Determine whether modified second-law statements can be recovered for restricted observables or under coarse-graining.
- Transport, currents, and hydrodynamics
- Extend the framework to explicitly account for energy/particle currents in macroscopic nonuniform operations and relate the results to hydrodynamic constraints and nonequilibrium steady states.
Practical Applications
Immediate Applications
The findings in the paper can lead to immediate applications in industry and academia, particularly in fields involving quantum computing and thermodynamic systems.
Industry
- Quantum Computing Optimization:
- Application of the principles derived from the emergence of the second law in quantum systems can enhance error correction algorithms and resource management in quantum computing, potentially leading to more stable and efficient quantum computers.
- Sector: Software, Quantum Computing
- Dependencies: Availability and precision of quantum control technologies.
Academia
- Educational Framework for Quantum Thermodynamics:
- Use the theoretical basis provided by the paper to develop new curricula that focus on quantum thermodynamics, aiding in better understanding of statistical mechanics at a quantum level.
- Sector: Education
- Dependencies: Integration with existing physics curricula and educational materials.
Long-Term Applications
The research paves the way for numerous long-term applications that could radically transform how quantum systems are integrated into various technological domains.
Industry
- Quantum-Thermodynamic Devices:
- Engineering devices that utilize the reconciliation of thermodynamics with pure quantum states for more efficient energy conversion systems.
- Sector: Energy, Engineering
- Dependencies: Significant technological advancements in controlling macroscopic quantum systems.
Policy
- Regulatory Standards for Quantum Technologies:
- Developing standards and frameworks based on the second law of thermodynamics in quantum systems to guide the ethical and effective deployment of quantum technologies.
- Sector: Governance, Policy
- Dependencies: Multidisciplinary collaboration among physicists, technologists, and policymakers.
Daily Life
- Quantum-Enhanced Consumer Electronics:
- Household electronics that leverage quantum states to optimize energy use, contributing to sustainable innovations.
- Sector: Consumer Electronics
- Dependencies: Mass production capabilities and consumer acceptance of quantum technologies.
Assumptions and Dependencies
The practical applications derived from this paper are contingent upon several key assumptions and dependencies:
- Technological Development: The advancement of quantum control technologies to implement theoretical principles in practical scenarios.
- Economic Viability: Cost-effectiveness of integrating quantum-thermodynamic principles into existing infrastructure.
- Academic and Industry Collaboration: Successful translation of research findings into market-ready technologies and policies requires ongoing collaboration between academia and industry.
- Policy Framework: Establishment of global standards and regulations to ensure safe and ethical use of quantum technologies.
Overall, the paper offers foundational insights that, upon further development and research, could lead to transformative applications across multiple fields and sectors.
Glossary
- Adiabatic thermodynamic operations: Thermodynamic operations in closed systems that exchange no heat with the environment, represented quantum mechanically via time-dependent Hamiltonians. "thermodynamic operations in systems that exchange no heat with the environment (not to be confused with the adiabatic theorem in quantum mechanics)."
- Additive Hamiltonian: A Hamiltonian that is a sum of local terms, corresponding to additive quantities, used to generate macroscopic operations. "We also introduce a macroscopic operation as unitary evolution generated by a time-dependent additive Hamiltonian, which is regarded as corresponding to adiabatic operations."
- Additive observable: An extensive observable built by summing translates of a fixed local operator over a macroscopic region; it corresponds to spatial averages of local quantities. "a quantum state, including pure states, is said to represent iMATE if the expectation values of all additive observables, which correspond to additive quantities in thermodynamics, agree with their equilibrium values."
- Canonical Gibbs state: The thermal equilibrium state of a system at inverse temperature β, given by ρ = e{−βH}/Z. "We say that a state ρ{L} represents an iMATE if ρ{L} is macroscopically equivalent to the canonical Gibbs state"
- Diagonal entropy: An entropy measure defined from the diagonal elements of the density matrix in the energy eigenbasis; it vanishes for energy eigenstates. "For instance, diagonal entropy~\cite{Santos2011,Ikeda2015Second} becomes zero when applied to energy eigenstates."
- Eigenstate Thermalization Hypothesis (ETH): The hypothesis that individual energy eigenstates (in the bulk of the spectrum) yield thermal expectation values for observables. "the eigenstate thermalization hypothesis (ETH)~\cite{Neumann1929,Deutsch1991,Srednicki1994,Rigol2008} states that every energy eigenstate located in the bulk of the energy spectrum represents thermal equilibrium"
- Extensive work: Work that scales proportionally with system size; thermodynamic second law constraints forbid extracting it in certain operations. "no extensive work can be extracted from any quantum state representing iMATE through any macroscopic operations with the operation times independent of the system size."
- Gibbs ensembles: Statistical ensembles describing thermal equilibrium (e.g., canonical ensemble) used in statistical mechanics. "it is widely believed that not only Gibbs ensembles but also other quantum states, including pure quantum states, can represent thermal equilibrium."
- Law of increasing entropy: A formulation of the second law stating that a closed system’s thermodynamic entropy cannot be decreased by adiabatic operations. "The law of increasing entropy states that thermodynamic entropy of a closed system cannot be decreased by any adiabatic operation."
- Lieb-Robinson bound: A bound that limits the speed of information propagation in quantum lattice systems, used to control locality in dynamics. "Using the Lieb-Robinson bound~\cite{Gong2020}, we show that macroscopic equivalence is preserved by any macroscopic operation within an operation time that is independent of "
- Long-time average: The time average of a state under unitary evolution over an infinite duration, used to define a relaxed final state. "We interpret the long-time average of the state at as the final state of the relaxation process"
- Macroscopic equivalence: An equivalence relation on states where all additive observable densities agree in the thermodynamic limit. "we say that two quantum states and are macroscopically equivalent, denoted by"
- Macroscopic operation: A unitary evolution driven by a time-dependent Hamiltonian that couples uniform external fields to additive observables, with operation time independent of system size. "We call such a unitary operation a macroscopic operation of operation time ."
- Macroscopic thermal equilibrium (MATE): A notion of equilibrium where macroscopic observables’ measurement outcomes almost surely match equilibrium values. "A quantum state is said to represent MATE if the measurement outcomes of macroscopic observables almost surely agree with their equilibrium values."
- Microscopic thermal equilibrium (MITE): A notion of equilibrium where microscopic observables’ expectation values match those in the Gibbs state. "The MITE, on the other hand, is characterized by the property that the expectation values of microscopic observables agree with those in the Gibbs state"
- Nonintegrable systems: Quantum many-body systems lacking a complete set of conserved quantities, typically expected to satisfy ETH. "and is believed to hold for typical nonintegrable systems"
- Operator norm: The norm of an operator given by its largest singular value; used to quantify the size (extensivity) of additive observables. "the operator norm~ of satisfies "
- Passivity: A property of states where no unitary can lower the energy expectation value; relates to impossibility of work extraction. "A quantum state is said to be passive for a given Hamiltonian if the expectation value of the Hamiltonian cannot be decreased by applying any unitary time evolution."
- Planck's principle: A second-law statement that energy of a closed system cannot be lowered by adiabatic operations with unchanged control parameters. "Here, Planck's principle states that the energy of a closed system cannot be decreased by any adiabatic thermodynamic operation in which the initial and final values of the control parameters, such as volume and magnetic field, coincide."
- Reduced density matrix: The density matrix of a subsystem obtained by tracing out the rest of the system; used to characterize local properties. "Consider the reduced density matrix $\mathrm{Tr}_{\Lambda_L\setminus\Cell[\bm{r}]}[\rho_L]$ on $\Cell[\bm{r}]$"
- Relaxation process: Time evolution under a fixed Hamiltonian after an operation, leading the system towards equilibrium-like behavior. "followed by a time-independent relaxation process."
- Thermodynamic limit: The limit of infinite system size used to define macroscopic quantities and equivalence of states. "When taking the thermodynamic limit , we consider such proper sequences of macroscopic subsystems"
- von Neumann entropy: Quantum entropy defined as S(ρ) = −Tr(ρ log ρ); it can fail to match thermodynamic entropy for pure states. "[Unfortunately, the von Neumann entropy does not work in general since it is zero for a pure quantum state even if the state represents thermal equilibrium and does not agree with thermodynamic entropy.]"
Collections
Sign up for free to add this paper to one or more collections.