- The paper demonstrates that bounded (Jacobi) and unbounded (CIR) models yield notably different tail risk predictions in SI epidemic dynamics.
- The authors derive explicit expressions for cumulative transmission intensity using Laplace and moment-generating techniques.
- Preliminary intervention strategies based on finite cumulative intensity highlight the critical role of model selection in epidemic control.
Modeling SI Epidemics with CIR and Jacobi Processes: Asymptotics and Interventions
This work investigates the incorporation of stochasticity into the time-varying transmission rate of the SI (Susceptible-Infected) epidemic model using two distinct positive processes: the Cox–Ingersoll–Ross (CIR) process and the Jacobi process. Traditional deterministic formulations with constant transmission do not capture observed epidemic variability, particularly volatility and tail-risk scenarios arising from environmental stochasticity and behavioral unpredictability. Here, the transmission rate βt is expressed as βt=φ(t)Pt, with φ(t) encoding deterministic intervention effects (e.g., public health responses) and Pt following either the CIR or Jacobi process, each ensuring positive sample paths but differing fundamentally in their support and tail behavior.
Under this framework, epidemic trajectories reduce to deterministic mappings of the stochastic cumulative transmission intensity Ht=∫0tβsds. The analysis centers on how the asymptotic and transient behaviors of the infected fraction, intervention efficacy, and risk quantification depend on the properties of this integrated intensity under different modeling choices for Pt.
Analytical Results: Stochastic Epidemic Dynamics
By explicitly solving the SI equation with stochastic time-dependent transmission, the infected and susceptible fractions at time t are given as transformations of Ht. The long-time behavior of the epidemic is dichotomous, depending on whether Ht remains finite or diverges. Two key theorems emerge:
- Absence of intervention (φ≡1): For ergodic Markov processes with stationary distribution having positive support and finite mean, βt=φ(t)Pt0 almost surely. Thus, the entire population is eventually infected (i.e., βt=φ(t)Pt1 almost surely).
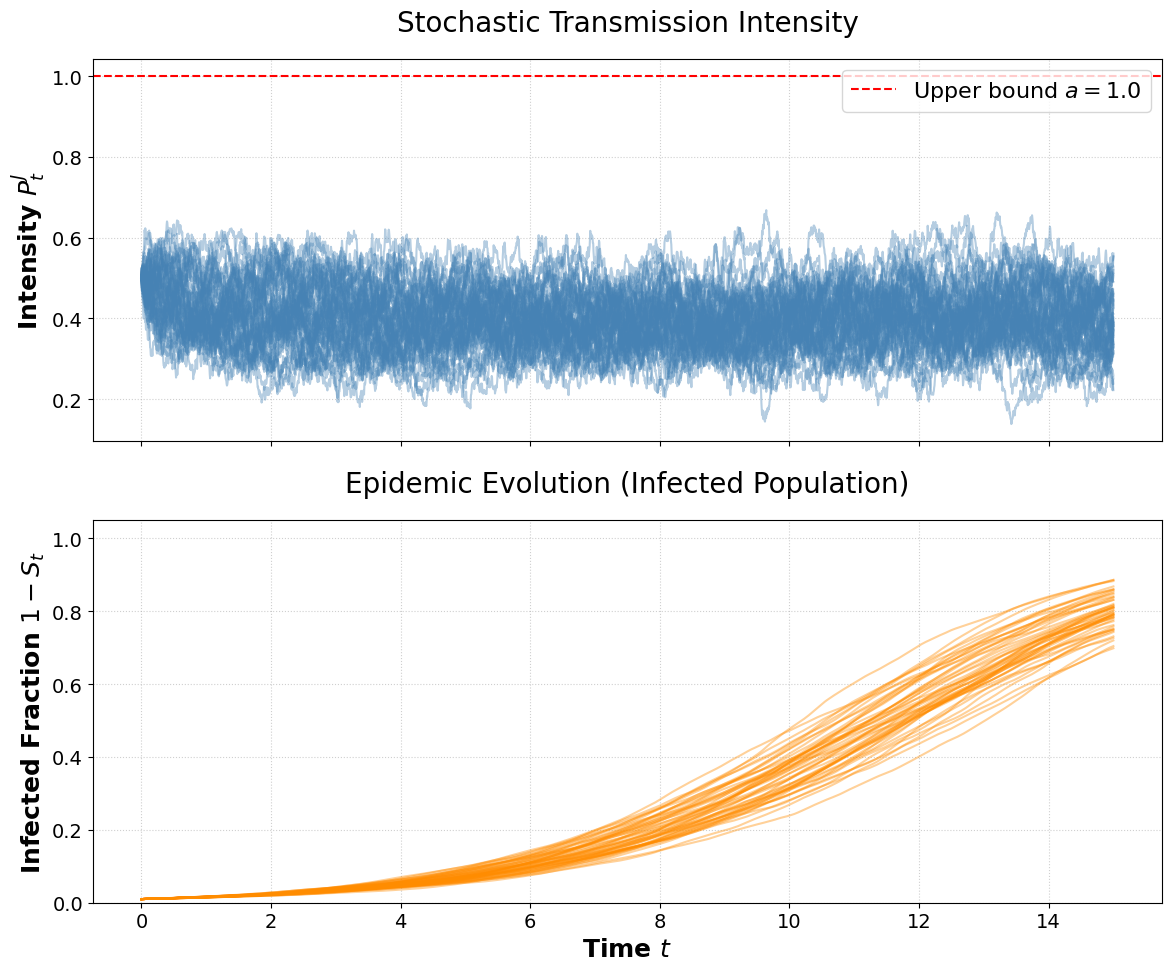
Figure 1: Stochastic sample paths for Jacobi-driven transmission (βt=φ(t)Pt2) illustrate inevitable epidemic saturation.
- Successful intervention (βt=φ(t)Pt3): For appropriately regular βt=φ(t)Pt4 (bounded mean), interventions ensure βt=φ(t)Pt5 is almost surely finite and a nonzero fraction of the population escapes infection. The limiting susceptible fraction remains strictly between zero and the initial state.
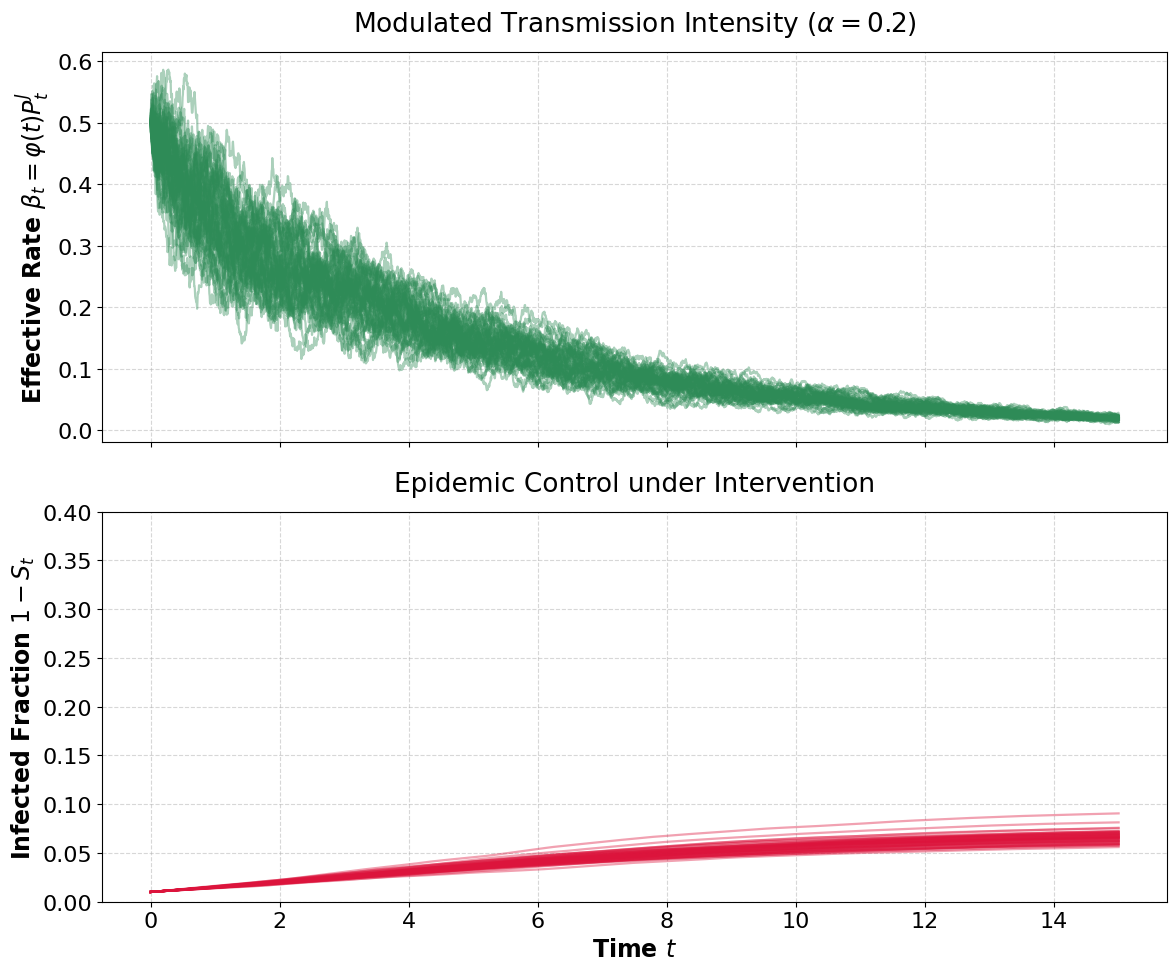
Figure 2: Exponential decay strategies modulate transmission and ensure disease saturation halts prior to complete infection.
The explicit equations governing the expected cumulative intensity are derived for both models:
- Jacobi: βt=φ(t)Pt6
- CIR: βt=φ(t)Pt7
These allow for direct verification of intervention sufficiency by controlling expectation bounds.
Model Specifics: Jacobi vs. CIR Dynamics
Jacobi Process
The Jacobi driver βt=φ(t)Pt8 is bounded in βt=φ(t)Pt9 and admits stationary Beta distributions under Feller-like parameter regimes. Its stochasticity reflects uncertainty in contacts per unit time but enforces strict limits corresponding to physical/social constraints.
CIR Process
The CIR driver φ(t)0 is non-negative but unbounded, with stationary Gamma distribution (when φ(t)1). While capturing mean-reverting properties, its unbounded support renders it structurally capable of arbitrarily large transmission bursts.
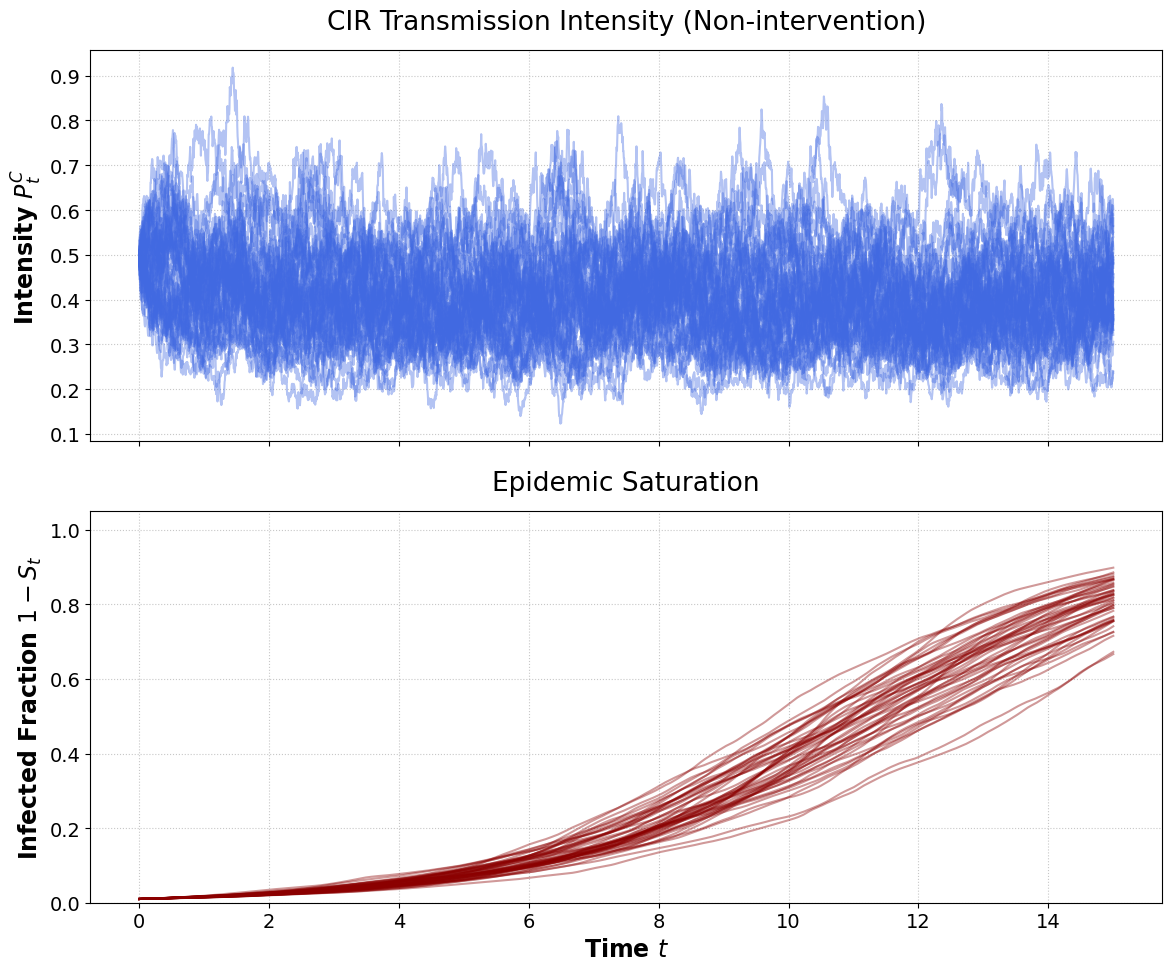
Figure 3: CIR transmission rate sample paths show substantial, unconstrained upward excursions compared to Jacobi dynamics.
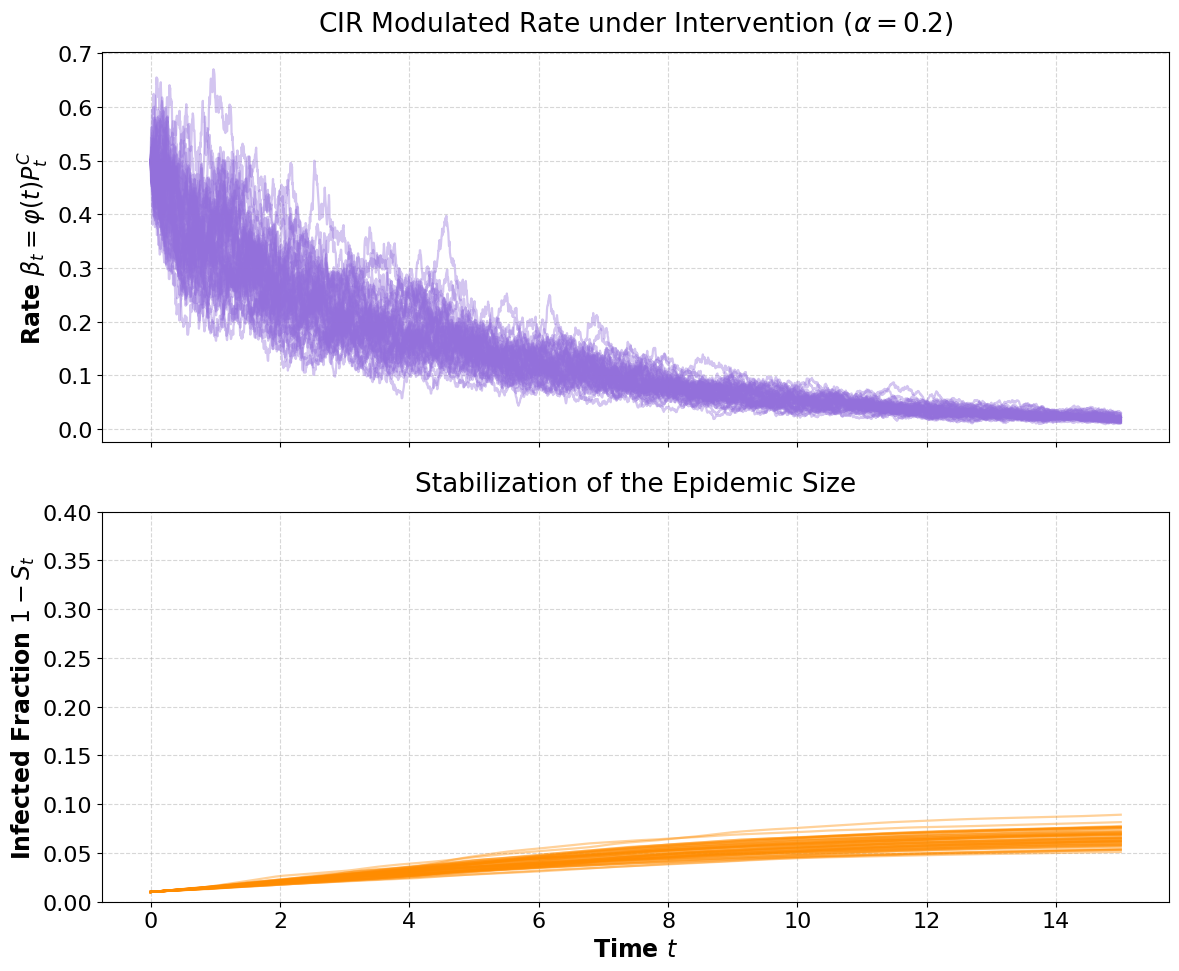
Figure 4: CIR-based infected fraction evolution with exponential intervention displays incomplete epidemic size, confirming intervention efficacy.
In the non-intervention regime, both processes inevitably drive the population toward full infection. However, their cumulative intensity distributions differ notably in the tails due to the CIR’s lack of an upper limit.
Risk Quantification and Thresholds
A critical practical question concerns the probability and timing of major outbreaks. The distribution of the random hitting time φ(t)2 to reach a given infection threshold is characterized via the distribution of φ(t)3. For the CIR process, the affine structure enables analytic Laplace and moment-generating functionals for φ(t)4, facilitating Chernoff-type upper bounds on φ(t)5:
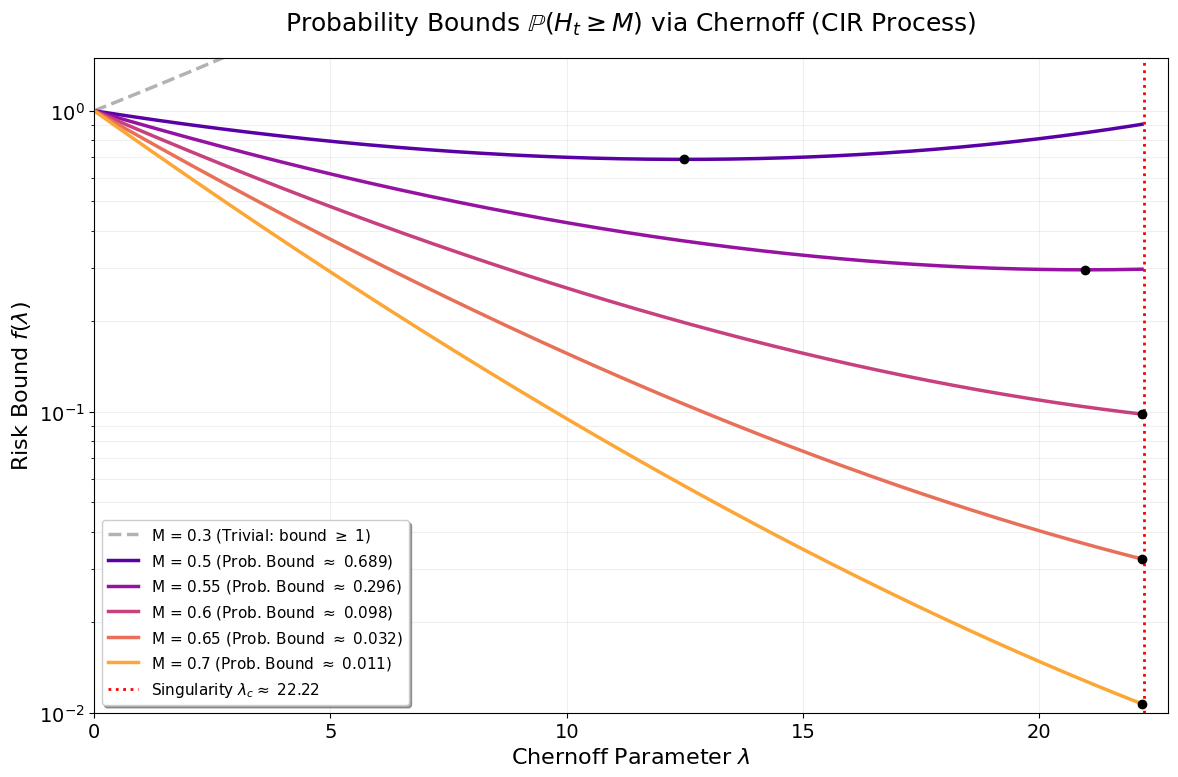
Figure 5: Numerical optimization of Chernoff risk bounds φ(t)6 for φ(t)7 as a function of threshold φ(t)8; higher thresholds yield sharper, lower bounds and shift the optimizing φ(t)9 towards the critical point.
This approach highlights the capacity for sharp risk assessment and tail-event estimation under different modeling assumptions.
Numerical Comparison: Implications of Model Choice
Central to the investigation is a systematic comparison between Jacobi and CIR frameworks when calibrated to identical stationary means and variances. The differences emerge predominantly in transient and extreme scenarios:
- Low-volatility regime: Both models behave equivalently, with infected and cumulative intensity distributions nearly identical.
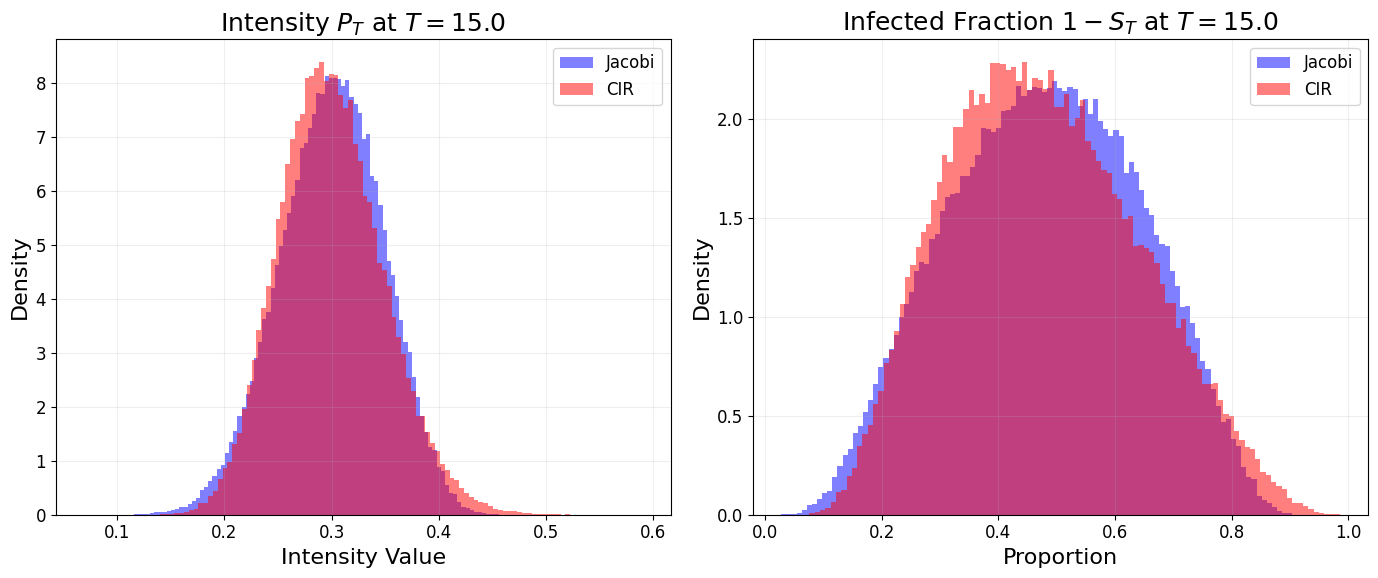
Figure 6: Under low volatility, Jacobi and CIR models yield indistinguishable infection intensity and epidemic size distributions.
- High-volatility or long-time horizons: The unbounded CIR process generates heavier right tails in both Pt0 and the infected fraction distribution. Extreme outbreaks (rare but high-impact) are much more probable under CIR.
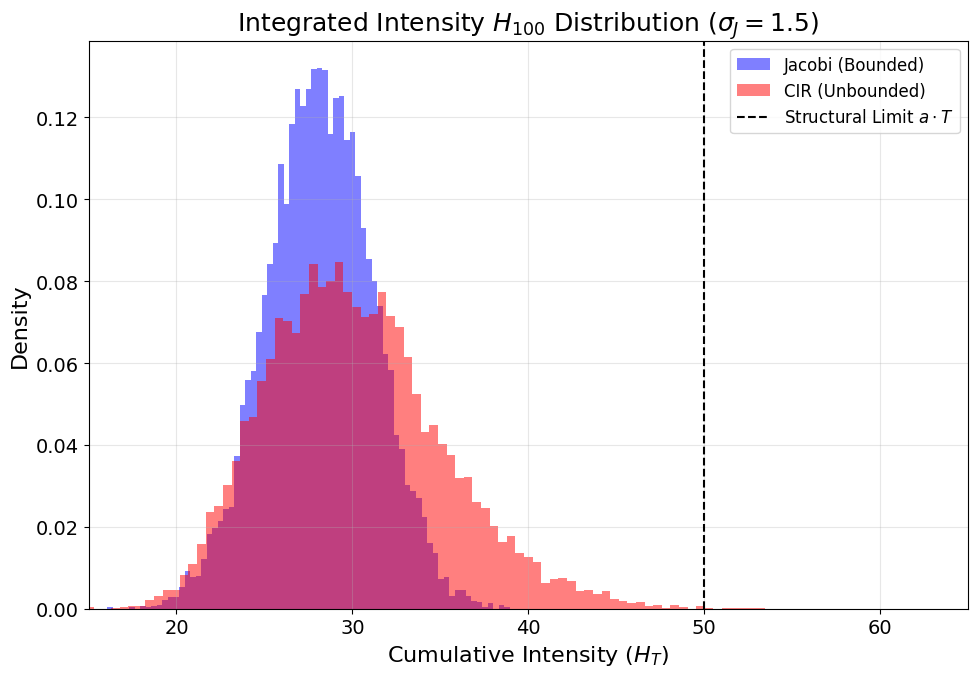
Figure 7: At long horizons, CIR supports extreme cumulative intensities impossible under bounded Jacobi, impacting risk estimates.
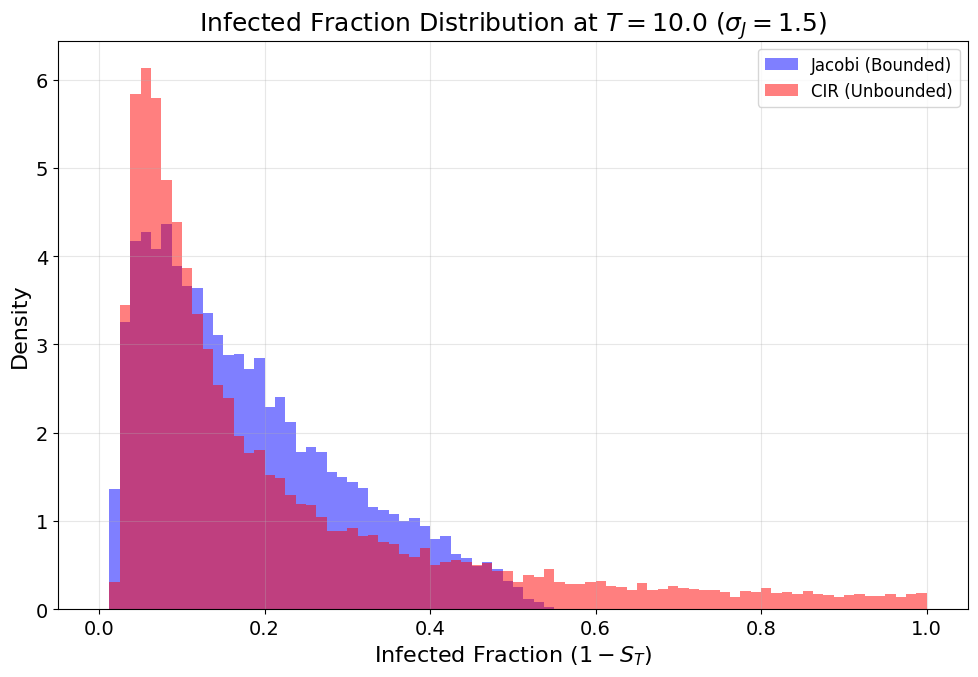
Figure 8: Early epidemic stages reveal a heavier right tail for CIR-driven infected fractions, indicating faster potential outbreak propagation.
- Even with strong interventions: The CIR model’s heavy tails persist, producing significantly larger “value-at-risk” estimates for epidemic size, despite matched first and second moments.
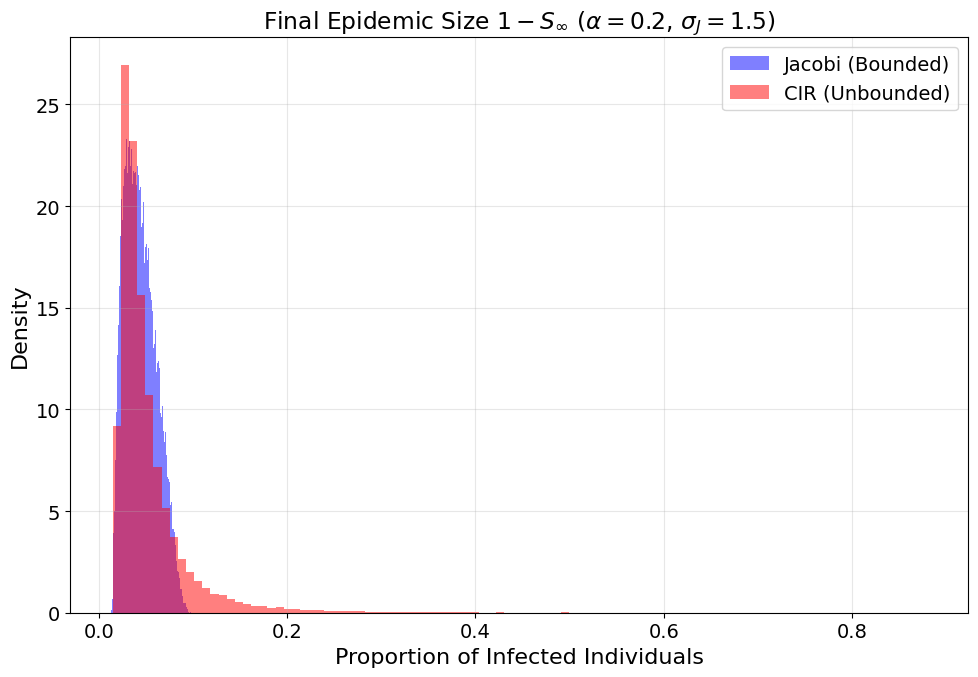
Figure 9: With exponential intervention, Jacobi’s bounded support sharply limits possible epidemic sizes; CIR allows rare but severe outbreaks.
A table of key risk metrics (omitted here for brevity) further quantifies this divergence, demonstrating that VaR and maximal observed outcomes are consistently and materially larger for CIR—implicating model selection as a critical consideration in public health risk estimation, particularly under volatile or poorly controlled transmission.
Theoretical and Practical Implications
The study exposes the non-robustness of epidemic risk quantification to the choice of stochastic transmission driver, even when calibrating standard moments. The bounded vs. unbounded support is the structural driver of tail risk and extreme event predictions. This result has several implications:
- Public Health Preparedness: Policies based on bounded models may systematically underestimate the risk and size of rare, severe outbreaks—potentially leading to insufficient mitigation resource allocation.
- Inference and Model Selection: The challenge of empirically distinguishing between bounded and unbounded transmission dynamics using epidemic time series is acute, especially as both models can fit mean behavior. Statistically consistent estimation and goodness-of-fit techniques sensitive to extremes are needed.
- Analytical tractability: The CIR process, through its affine structure, supports closed-form Laplace and moment-generating functional computation for cumulative intensity. This enables rigorous risk quantification and optimization-based scenario assessment.
- Future Directions: The paper identifies several open problems, including nonparametric estimation of Pt1 jointly with Pt2 from data, and further theoretical work on explicit threshold solutions for intervention functions outside the exponential class.
Conclusion
This work rigorously demonstrates the critical role of the structural properties of stochastic transmission processes in SI epidemic models. Bounded (Jacobi) and unbounded (CIR) models, though statistically similar at low volatility or under strong mean reversion, yield dramatically different predictions for extreme epidemic outcomes and intervention efficacy in volatile scenarios. The analytic and computational machinery developed around the CIR process provides a robust framework for risk-sensitive public health planning—while also highlighting the necessity of careful model selection and calibration when forecasting with stochastic epidemiological models.
For further technical details and theoretical development, see "Modeling Transmission Intensity in SI Epidemics via CIR and Jacobi Processes: Asymptotic Results and Preliminary Intervention Strategies" (2604.02224).








