- The paper establishes a thermodynamic framework that quantifies noble-gas solubility in both solid and liquid metallic hydrogen at 500 GPa.
- It employs AIMD simulations and first-principles free-energy calculations to reveal phase-dependent impurity energetics and local lattice distortions.
- The results explain atmospheric noble-gas partitioning in giant planets by highlighting the stabilizing effect for heavier species in the liquid phase.
Introduction
The thermodynamic stability of trace impurities in metallic hydrogen critically informs models of planetary composition and differentiation, with noble gases (He, Ne, Ar, Kr, Xe) exhibiting anomalous partitioning in giant planet atmospheres. This study establishes a quantitative thermodynamic framework for noble-gas solubility in both solid and liquid metallic hydrogen at 500 GPa, employing ab initio molecular dynamics and first-principles free-energy calculations. The formalism enables explicit comparison of substitutional energetics, structural signatures, and the phase dependence of impurity incorporation, revealing mechanisms underlying noble-gas fractionation in planetary interiors.
Methodological Framework
State-of-the-art AIMD simulations combine the CASTEP code for finite-temperature sampling of microstates and DFT-based force calculations with quantum mechanical accuracy. The solid phase (crystalline I41/amd) is treated via NPT ensemble dynamics and subsequent DFPT phonon analysis, capturing both enthalpic and vibrational free-energy components. The liquid simulations employ NPT dynamics at 600 K to access the disordered metallic state prevalent deep within gas giants.
Substitutional impurity models systematically replace one or more hydrogen atoms with a noble gas, with formation free energy decomposed into electronic (static), vibrational (ZPE and Svib), and configurational (Sconf) terms. This approach rigorously distinguishes local impurity effects from collective phase behavior.
Results: Solid Solution Thermodynamics and Structural Analysis
Substitution and Local Distortion in the Solid Phase
Noble-gas incorporation is characterized by enlarged substitutional cavities in the hydrogen framework, scaling with atomic number. Despite substantial local distortion, the hydrogen network maintains the body-centered tetragonal I41/amd structure across all impurity types, with negligible reconstruction outside immediate impurity vicinity.
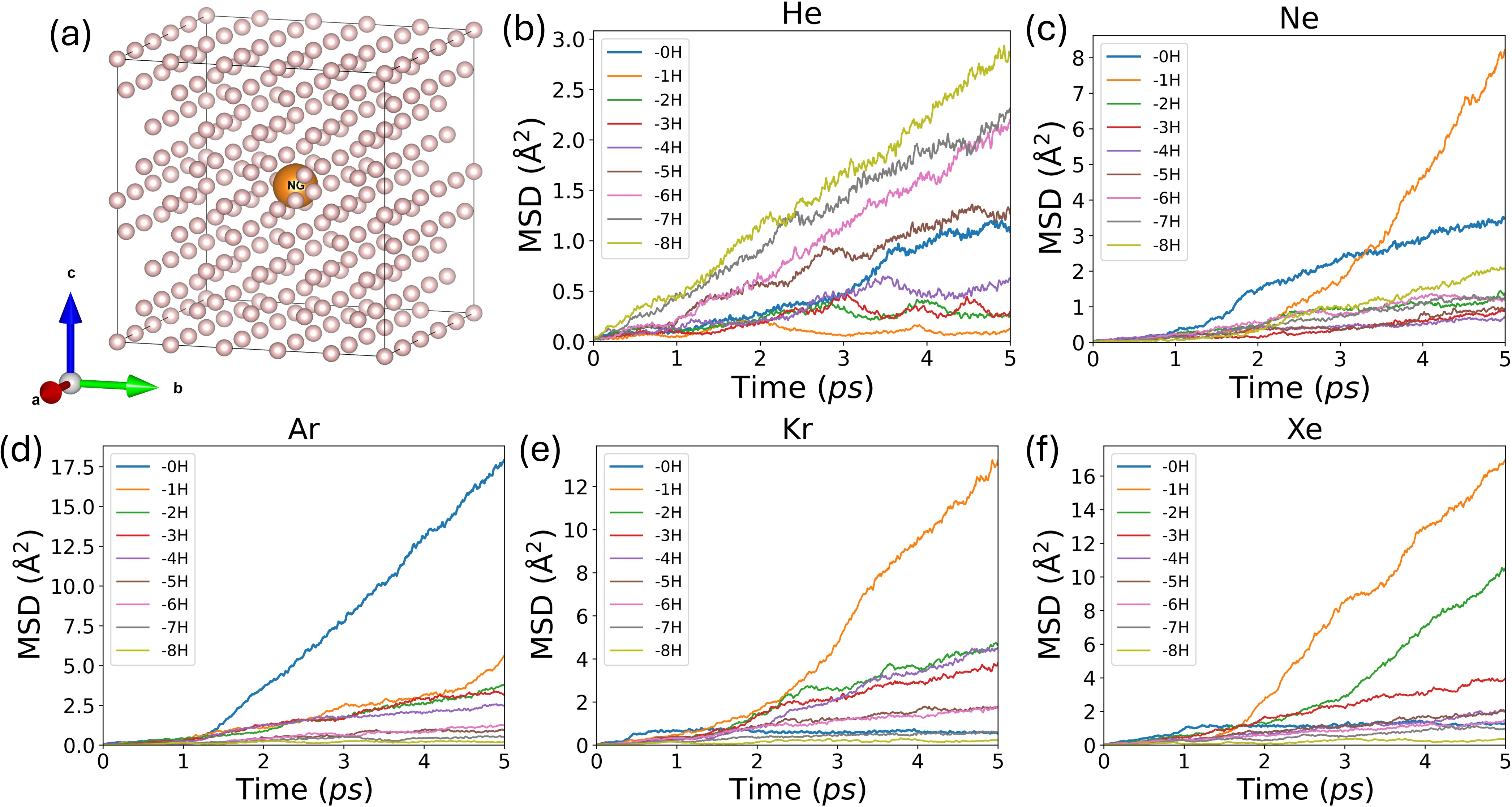
Figure 1: Substitutional noble-gas structures and time evolution of atom-averaged MSDs, indicating stability thresholds for each impurity species.
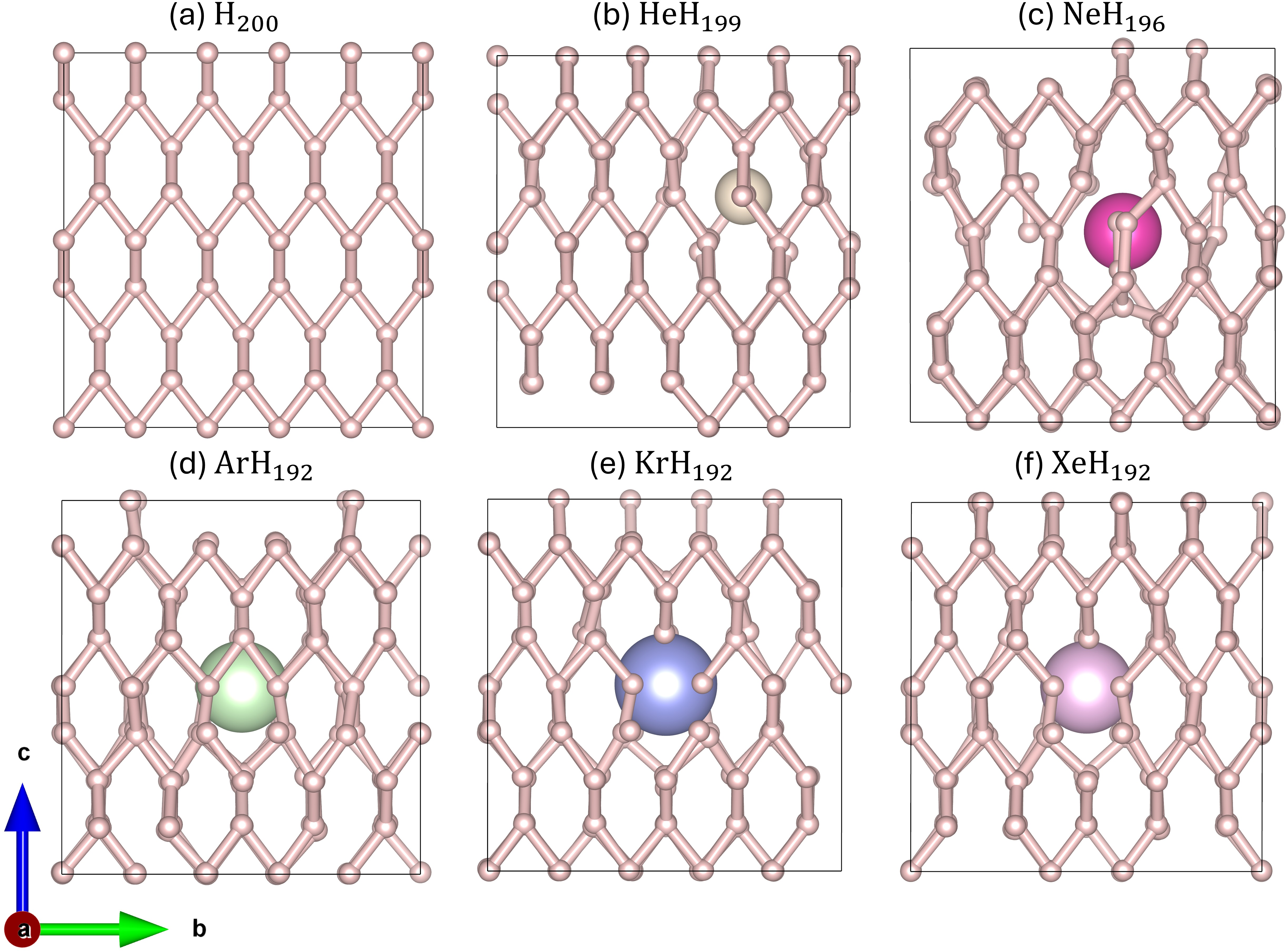
Figure 2: Optimized atomic configurations after AIMD, illustrating the localized lattice distortion imposed by He, Ne, Ar, Kr, and Xe compared to pristine H200.
Crucially, COHP analysis demonstrates minimal covalent interaction between noble gases and the hydrogen lattice, reflected in weak (negative) IpCOHP values. Vibrational density-of-states analysis confirms the absence of high-frequency vibrational modes corresponding to hydrogen-noble-gas bonding, with the exception of Ne, which induces intermediate-frequency stretching features.
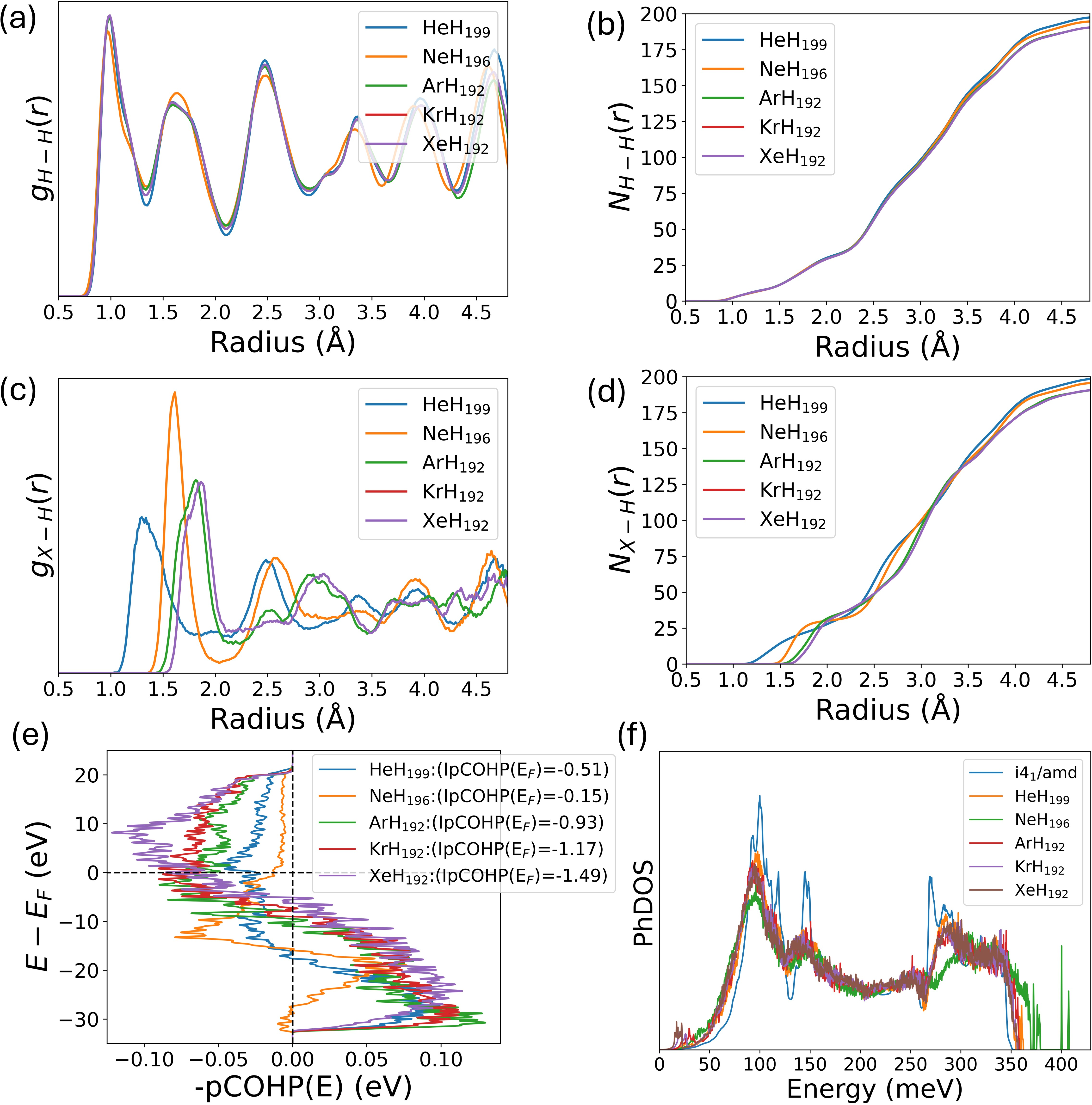
Figure 3: Radial distribution and cumulative coordination functions for H-H and impurity-H pairs, alongside COHP and phonon DOS, highlighting weak chemical interaction and local vibrational effects.
All noble-gas impurities exhibit strongly positive formation free energies in the solid phase (e.g., gHe=6.29 eV/cell, gNe=9.64 eV/cell, gAr=5.87 eV/cell at 500 GPa). The dominant unfavorable component is the electronic enthalpy—primarily a consequence of Pauli repulsion and lack of hybridization—further augmented by positive ZPE shifts due to local mode stiffening. The marginally favorable mixing and vibrational entropy are negligible relative to the enthalpic penalty. No noble gas forms a stable solid solution in metallic hydrogen under these conditions.
Results: Liquid Phase Solubility
Structural and Dynamical Signatures
In the liquid state, hydrogen exhibits canonical diffusive dynamics, while noble-gas species display reduced MSD slopes in proportion to atomic mass. The fluid structure, as evidenced by RDFs and coordination numbers, is robust to impurity insertion: the principal H-H peak is unaffected, and only subtle broadening is observed for heavier noble gases.
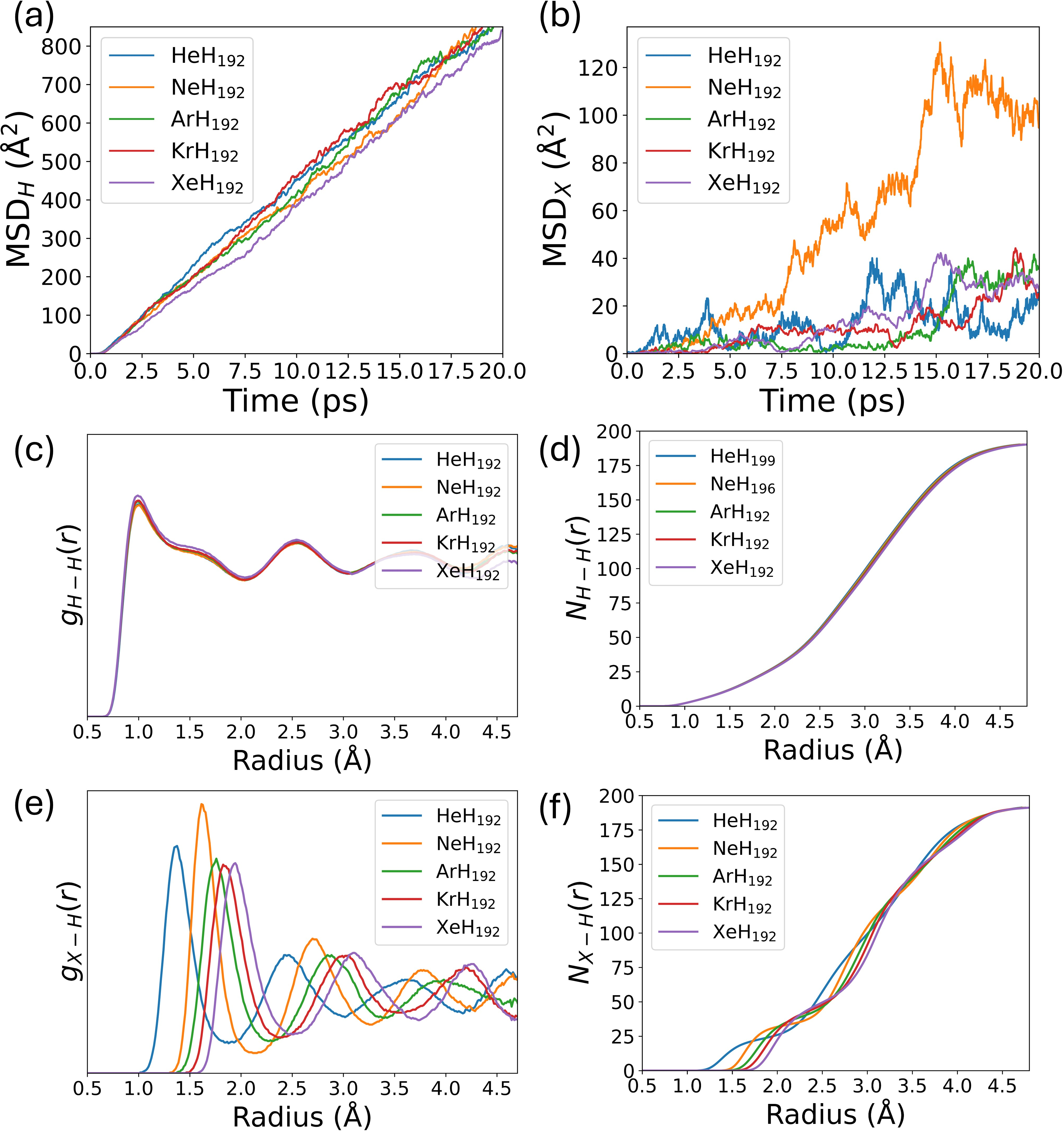
Figure 4: MSD comparison for H and noble-gas species, alongside liquid-phase structure correlations, demonstrating homogeneous hydrogen networks with locally perturbed impurity environments.
Enthalpy of Solvation
A salient finding is the systematic crossover in formation enthalpy for noble-gas impurities:
- He and Ne remain thermodynamically unstable (dHHe=0.14±0.15 eV/cell, dHNe=0.53±0.13 eV/cell).
- In contrast, Ar, Kr, and Xe display negative formation enthalpies (Svib0 eV/cell, Svib1 eV/cell, Svib2 eV/cell), indicating stabilization and solubility in the liquid phase.
This increased solubility with rising atomic number is attributable to the disorder-driven stabilization in the liquid, where the energy penalty for excluded volume and weak impurity-hydrogen coupling is partially offset by the entropic and configurational flexibility of the disordered network. The sharp contrast with the solid phase underscores critical phase dependence.
Discussion: Implications for Planetary Interiors and Noble-Gas Partitioning
The selective solubility of heavy noble gases in liquid metallic hydrogen has direct implications for planetary differentiation. In giant planet interiors, where metallic hydrogen is predominantly liquid, Ar, Kr, and Xe are stably retained within the metallic envelope, explaining observed atmospheric excesses. Conversely, He and Ne are excluded even from the liquid phase, supporting ongoing atmospheric depletion via "helium/neon rain"—an episodic precipitation rather than continuous rain, contrary to some simplified depictions.
The mechanistic insights provided—local impurity energetics, negligible covalent binding, and phase-driven entropy stabilization—point to the necessity of explicit phase-state modeling in interior structure calculations and core erasure processes. These results rationalize core erosion phenomena and the existence of compositional gradients (fuzzy cores) in the context of high-pressure geochemistry. They also clarify why noble-gas partitioning patterns differ qualitatively from models based solely on chemical inertness at ambient conditions.
This study further highlights the limitations of extrapolating from behavior in molecular hydrogen: in molecular hydrogen, all noble gases are soluble, and crystalline hydrates are stable, whereas in the metallic regime, strong selectivity emerges. Future high-pressure data sets and exoplanetary atmosphere observations should be reanalyzed in light of these findings.
Conclusion
A unified thermodynamic and atomistic exploration of noble-gas impurities in solid and liquid metallic hydrogen reveals an intrinsic phase dependence: all noble gases are insoluble in crystalline metallic hydrogen, while heavier species (Ar, Kr, Xe) are stabilized in the liquid phase. The origin of this behavior lies in the interplay between Pauli exclusion-driven enthalpic penalties and disorder-enabled entropic stabilization, with negligible chemical bonding. These results elucidate the microscopic basis for noble-gas fractionation in giant-planet interiors and provide key parameters for next-generation models of planetary evolution, core erosion, and atmospheric composition. Phase-specific impurity behavior must be incorporated explicitly in planetary structure and atmospheric transport modeling to achieve quantitative accuracy.



