- The paper establishes that CO and N₂ can be efficiently produced via photolysis and radiolysis of cometary ice analogs, with quantitative yields measured through IR spectroscopy and TPD.
- It demonstrates that matrix effects, such as water-induced cage constraints, notably reduce N₂ formation while modestly influencing CO production.
- Temperature and irradiation type modulate the yields, linking experimental outcomes to cometary observations and highlighting the need for low-temperature CO entrapment.
Experimental Design and Methodology
The authors systematically explore the photolytic and radiolytic formation of CO and N₂ from protostellar cometary ice analogs comprising H₂O, CO₂, and NH₃. Laboratory simulations were executed at SPACECAT and SPACETIGER, using UHV cryostats, calibrated UV lamps (peak λ ≈ 161 nm, flux ≈ 10¹⁴ cm⁻² s⁻¹) and 2 keV electron sources. Films were prepared with isotopically labeled CO₂ (13CO₂) and NH₃ (15NH₃) to unambiguously resolve daughter species (CO, N₂) amidst background. Compositions covered pure (primary), binary (H₂O:CO₂ or H₂O:NH₃), and ternary (H₂O:CO₂:NH₃) configurations, with astrophysically relevant ratios (e.g., 100:20:5). Irradiations spanned 10–100 K, targeting steady-state, with total photon/electron fluences imparted analogous to molecular cloud/outer disk exposures.
Quantification utilized IR spectroscopy (transmission/reflection) and Temperature Programmed Desorption (TPD) with QMS calibration to extract column densities of reactants and products. The destruction/formation kinetics were modeled by exponential fits, extracting cross sections and steady state concentrations. Band strength uncertainties (~20%) and experimental variability were rigorously propagated.
Mechanisms and Quantitative Conversion Efficiencies
Upon irradiation, CO₂ is efficiently converted to CO, and NH₃ to N₂ via photolysis and radiolysis. Pure ices exhibited high yields, with CO/CO₂(f) ratios of 51–62% and N₂/NH₃(f) of 15–21%. In water-rich binary and ternary analogs, CO yields reduced to 3–10% (relative to CO₂), and N₂ yields ranged 4–19% (relative to NH₃), with branching ratios reflecting matrix effects. Notably, CO formation from consumed CO₂ was nearly invariant across matrix complexity, whereas N₂ formation from NH₃ diminished with increasing water content, indicative of matrix-induced cage effects and hindered nitrogen fragment diffusion.
Irradiation temperature modulated yields: in ternary ices, conversion efficiencies for both CO and N₂ increased with T, with a noticeable minimum at 30 K attributed to volatility-induced product loss. Both UV and electron irradiation drove similar qualitative trends; however, electron bombardment gave higher N₂ yields in water-rich matrices, suggesting enhanced fragmentation favoring smaller nitrogenous moieties.
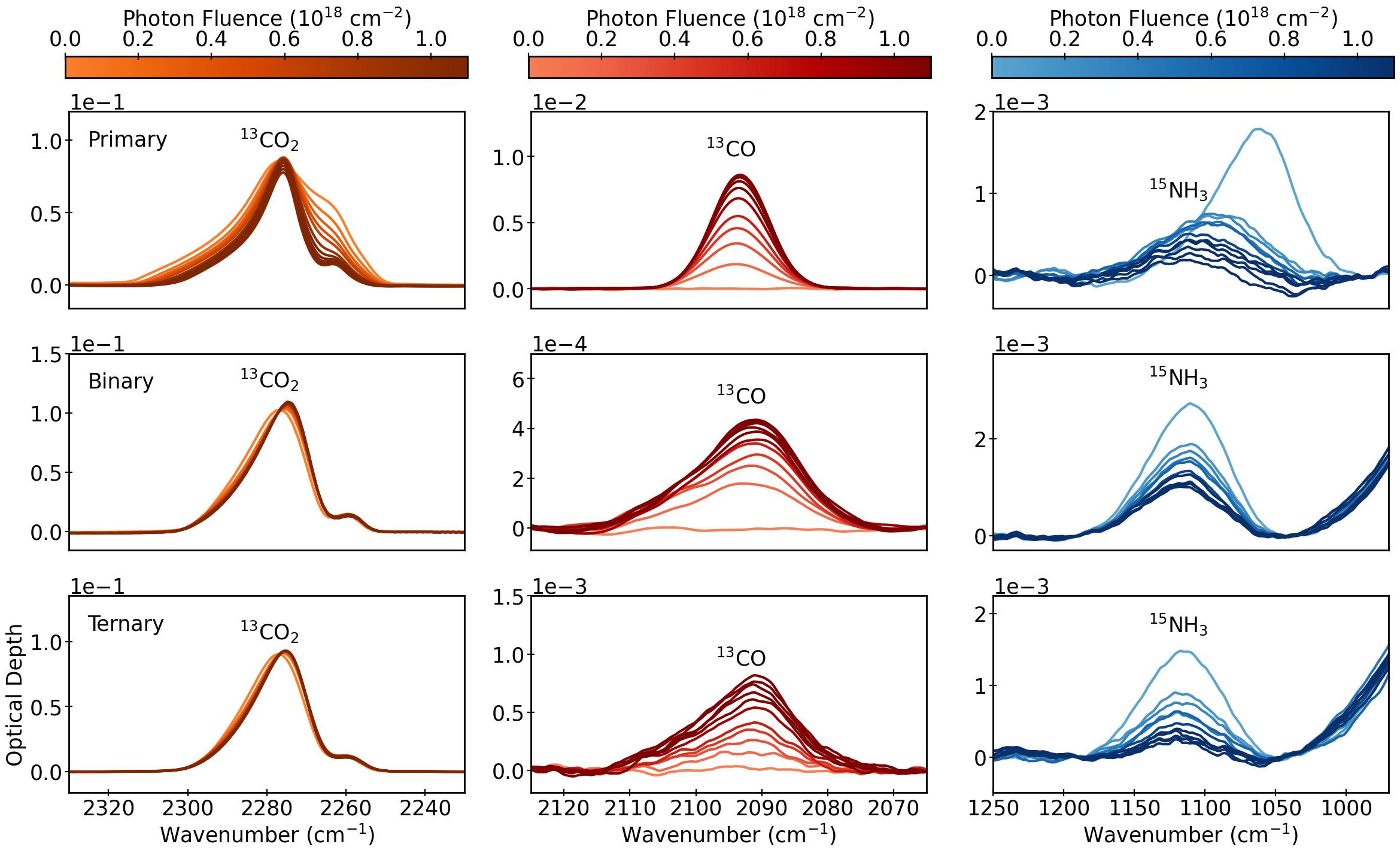
Figure 1: The infrared spectra of various ices throughout UV photolysis at 10 K, illustrating CO₂ consumption and CO, NH₃ transformations across pure, binary, and ternary matrices.
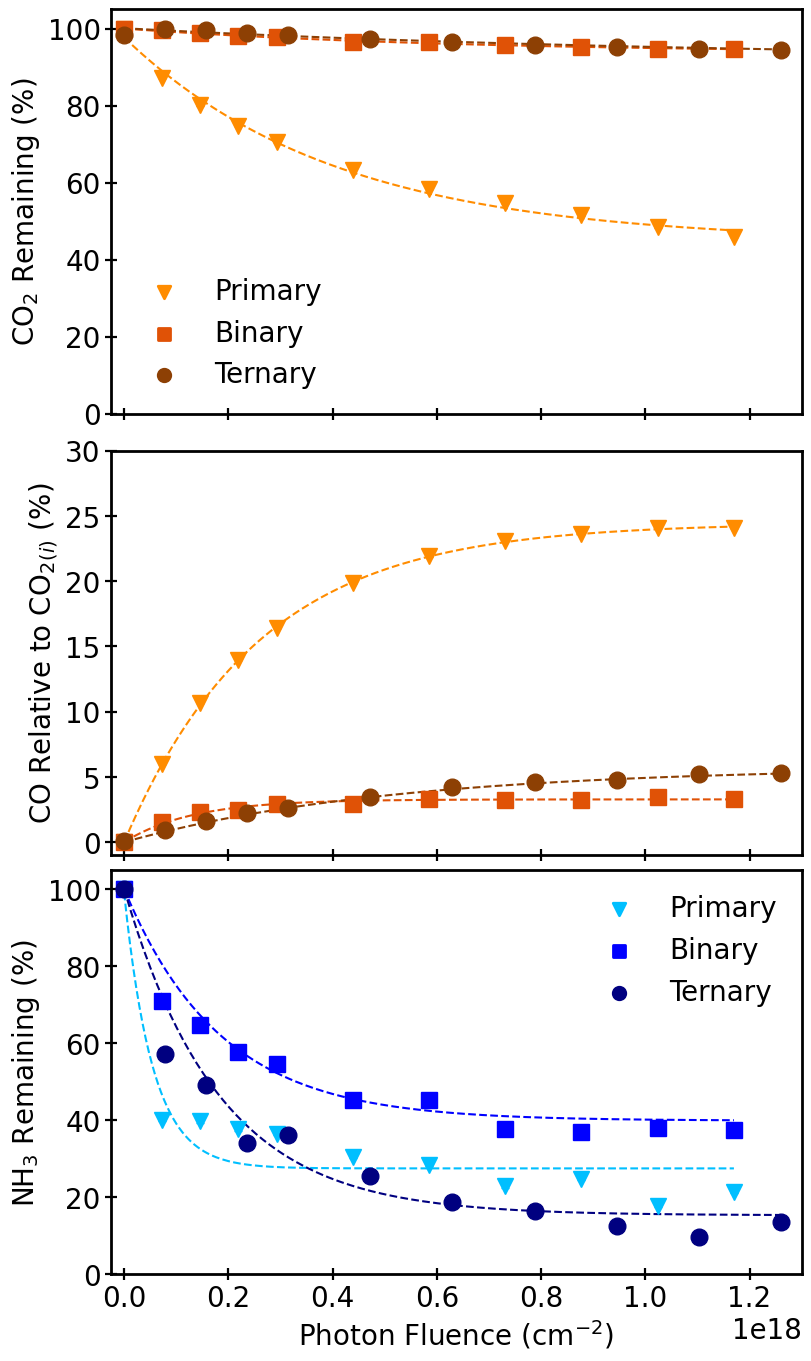
Figure 2: The temporal behavior of CO₂, CO, and NH₃ column densities during UV irradiation at 10 K reveals rapid initial conversion followed by plateauing at steady-state.
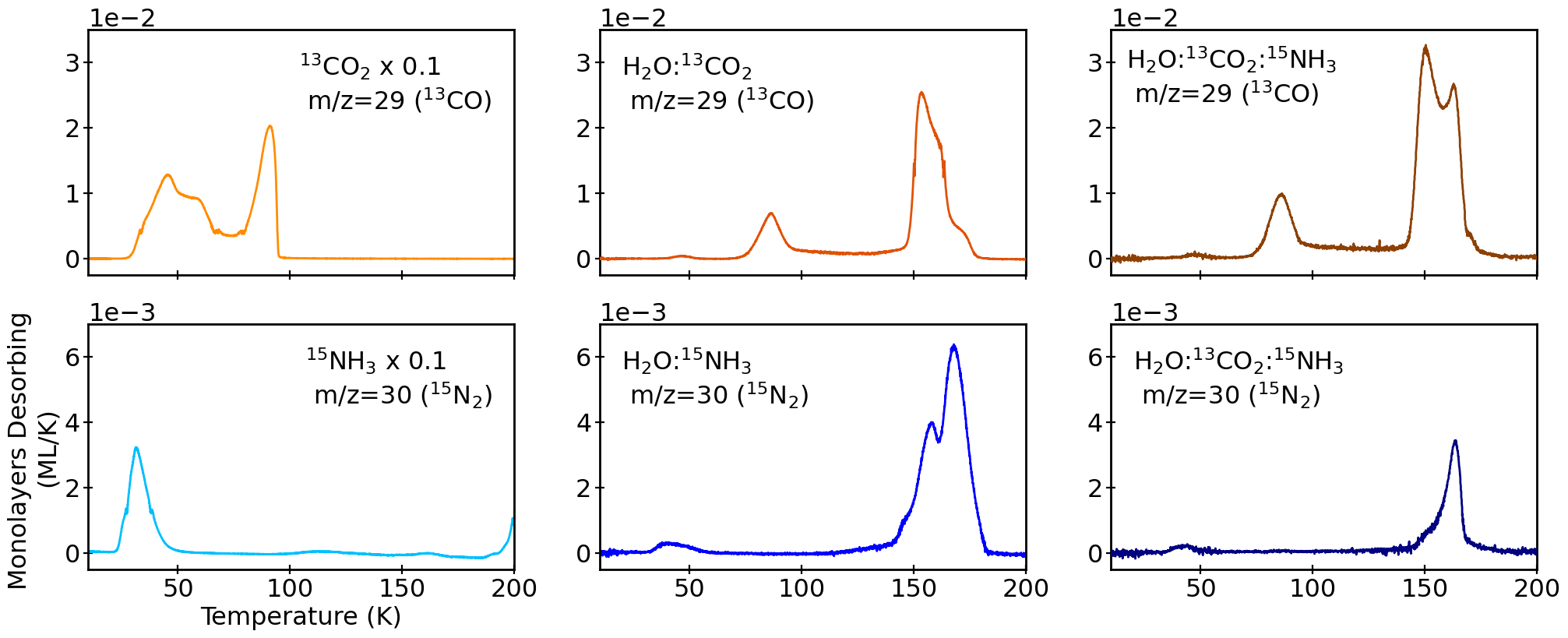
Figure 3: TPD curves for 13CO and 15N₂ in different matrices demonstrate distinct desorption behaviors, with matrix-dependent entrapment and release profiles.
Comparison to Comet Observations and Implications for Hypervolatile Origins
Experimental CO and N₂ abundances were compared to cometary measurements (e.g., 67P, Hale-Bopp, Hyakutake) relative to H₂O and parent volatiles, using CO/H₂O, CO/CO₂, N₂/H₂O, and N₂/NH₃ ratios.
- CO: Most comets exhibit CO/H₂O ratios (up to ~32%) that substantially exceed the maximum laboratory photolytic/radiolytic yields (0.9%). Only CO-poor comets (e.g., 103P, 73P) can be explained by in-situ CO₂ dissociation. Higher CO levels necessitate entrapment of gas-phase CO at sub-30 K, consistent with prior models.
- N₂: Cometary N₂/H₂O ratios (0.03–0.7%) and N₂/NH₃ ratios (up to ~19%) align closely with experimentally generated N₂ in water-rich matrices. Isotopic evidence from 67P (14N/15N ≈ 130 in both N₂ and NH₃) supports a NH₃-derived origin of cometary N₂, contrasting with the nebular value (~440), and rules out exclusive gas-phase inheritance for most comets.
- Temperature Dependence: Elevated laboratory yields at higher irradiation temperatures mirror a plausible scenario of icy grain processing in disks, supporting the notion that some hypervolatiles are produced and retained during grain evolution, subject to efficient entrapment mechanisms.
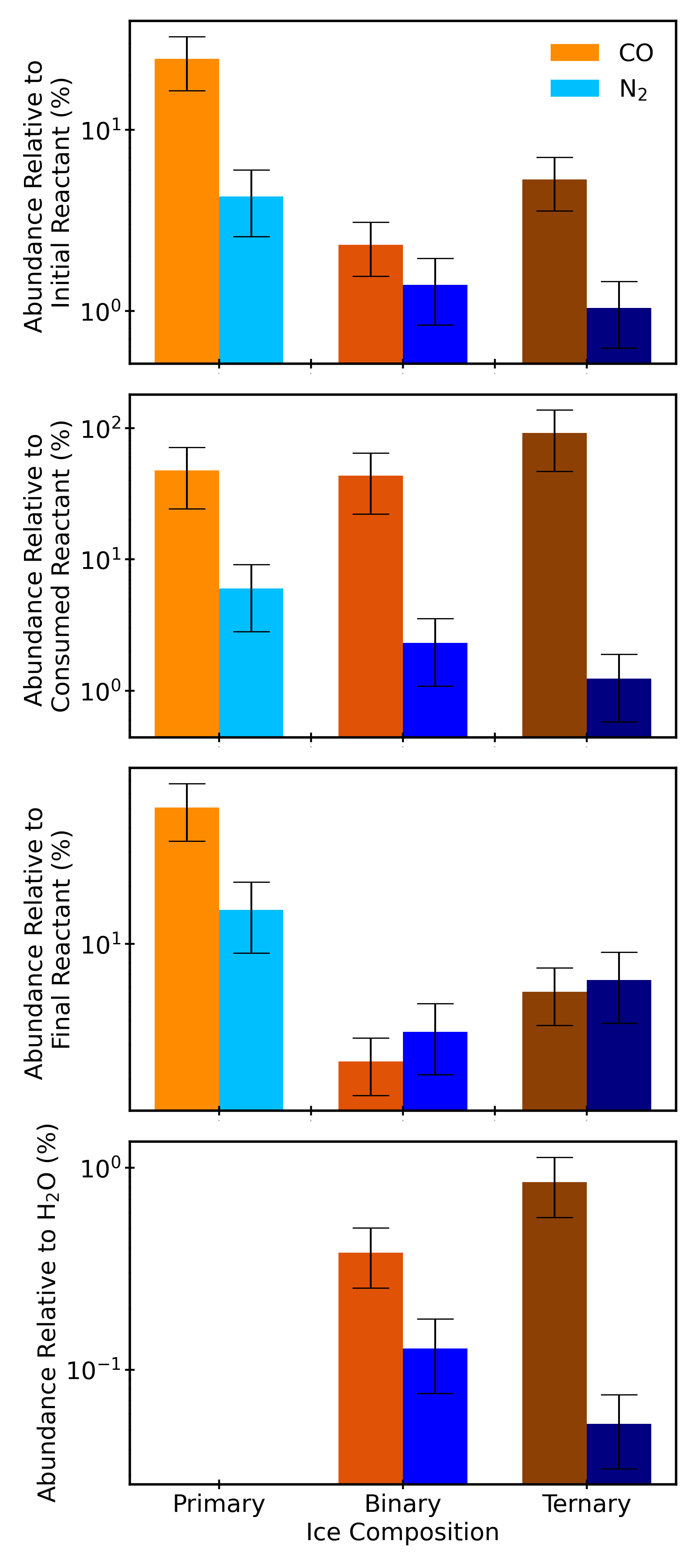
Figure 4: Summary plot of CO and N₂ yields across matrix types and irradiation conditions, highlighting the drastic reduction in hypervolatile formation in water-rich matrices and the increased N₂ yield by electron bombardment.
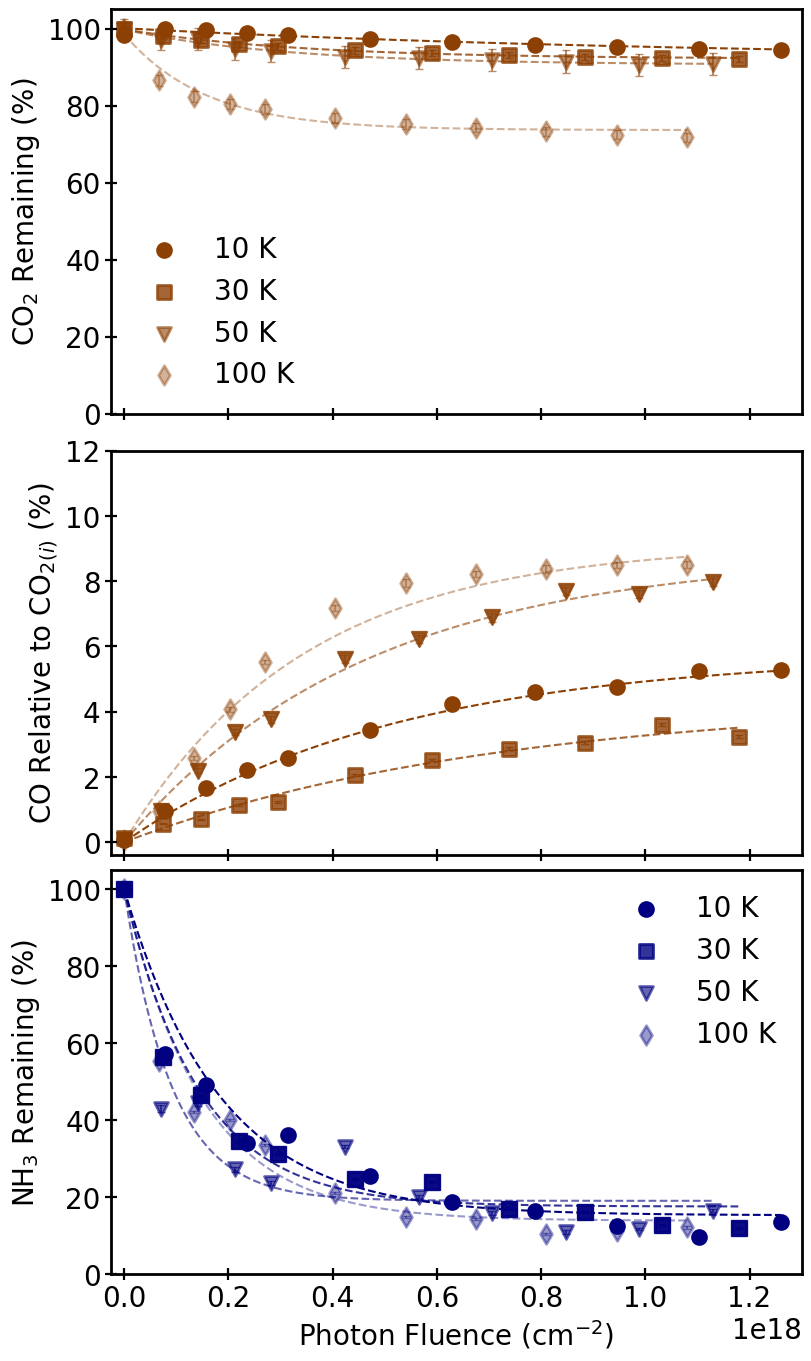
Figure 5: Comparison of CO and N₂ mixing ratios observed in comets versus those achieved in laboratory analogs. Cometary N₂/H₂O is fully accounted for by photoprocessing, while CO/H₂O generally exceeds laboratory production, requiring gas-phase entrapment.
The study challenges the canonical assumption that hypervolatile abundances in comets directly trace low-temperature formation conditions. Experimental results demonstrate that:
- N₂ formation via NH₃ photolysis/radiolysis in interstellar and disk ice analogs suffices to explain observed cometary N₂ abundances, thus N₂/H₂O ratios <1% should not be considered stringent formation temperature diagnostics.
- CO synthesis through CO₂ dissociation is quantitatively inadequate for most comets; major CO reservoirs demand entrapment of gas-phase CO below its sublimation temperature.
- Matrix effects decisively modulate photoproduct yields, with water ice imposing cage constraints on diffusion and recombination, disproportionately affecting N₂ formation pathways.
- Isotopic ratios corroborate photochemical processing for N₂ but cannot decisively distinguish origins of CO due to ISM precursor relationships.
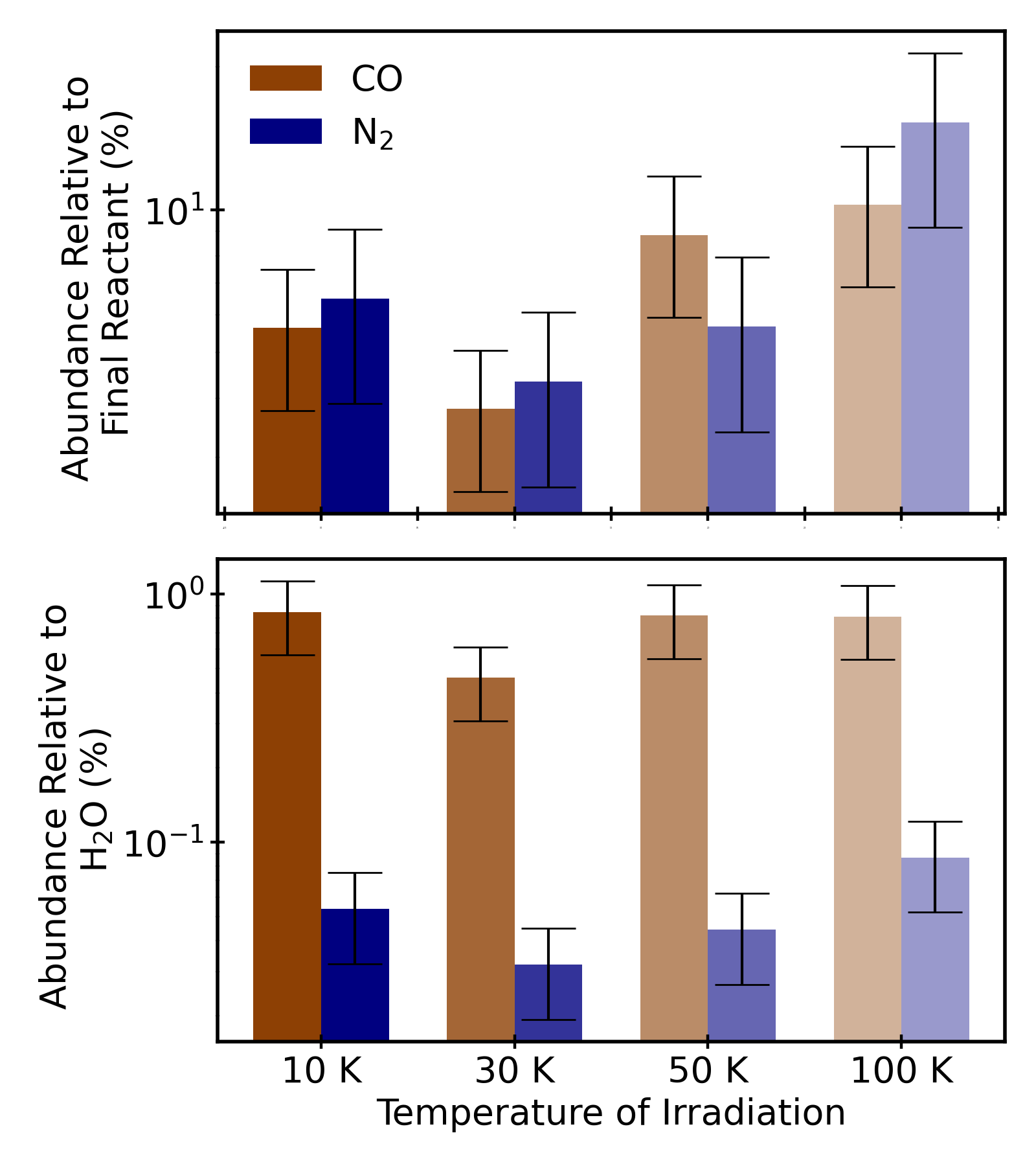
Figure 6: Histogram summarizing CO and N₂ yields as a function of irradiation temperature in ternary mixtures, showing increased hypervolatile formation at higher temperatures, except for product loss near volatile thresholds.
Future Directions
Further research is warranted to:
- Systematically assess the interplay of grain size, porosity, and matrix composition on photolytic/radiolytic yields and product entrapment.
- Expand isotopic analyses to other cometary volatile pairs to constrain their origins mechanistically.
- Develop integrated models coupling laboratory yields, migration-driven grain evolution, and observational constraints to delineate the chronology of hypervolatile reservoirs in planet-forming disks.
Conclusion
Laboratory irradiation experiments of cometary ice analogs establish robust quantitative upper bounds for CO and N₂ formation via photodissociation and radiolysis of CO₂ and NH₃. The results demonstrate that while photoprocessing can account for cometary N₂ abundances, especially with isotopic parity to NH₃, most observed cometary CO requires low-temperature entrapment. These findings necessitate caution in using hypervolatile ratios—specifically N₂/H₂O—as formation temperature proxies and compel a reevaluation of cometary chemical evolution paradigms. The work bridges detailed mechanistic laboratory insights with the interpretation of volatile inventories in Solar System and extrasolar comets, further informing models of planetesimal and planet formation.





