- The paper demonstrates that minute, sub-ångström shifts in the RPE65 active site lead to an exponential decline in proton tunneling activity.
- It employs a hybrid quantum-classical pipeline integrating AlphaFold predictions with VQE quantum simulations to model enzymatic thresholds.
- Findings establish a strong correlation between the Relative Quantum Activity Score and clinical severity, offering a new framework for variant stratification.
Introduction
This study rigorously investigates the biophysical consequences of pathogenic mutations in the human visual isomerase RPE65, the rate-limiting enzyme in the regeneration of 11-cis-retinal in the visual cycle. The central assertion is that clinical severity in RPE65-mediated retinal disease is not simply determined by macroscopic disruption of protein structure or classical enzymatic kinetics. Instead, the work demonstrates that the enzyme operates close to a quantum-critical regime, where minimal sub-ångström changes in active-site geometry precipitate an exponentially catastrophic decrease in enzymatic activity via suppression of proton tunneling. This threshold effect is termed the "Quantum Cliff." The paper presents a hybrid quantum-classical computational pipeline that integrates AlphaFold-predicted structures, ab initio quantum simulation using the Variational Quantum Eigensolver (VQE), and a mechanistically interpretable Relative Quantum Activity Score (RQAS) as a predictive metric for clinical stratification.
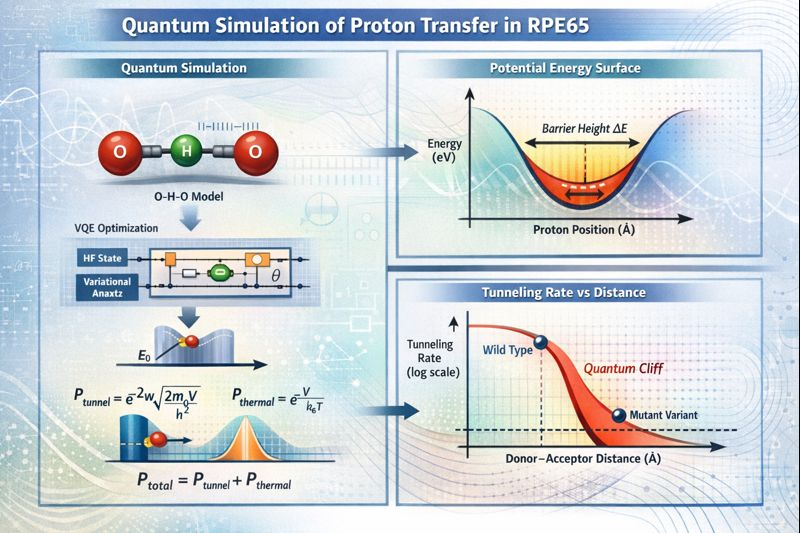
Figure 1: The RPE65 proton-transfer analysis pipeline combines AlphaFold-predicted structure with VQE quantum simulation of the O-H-O active site and a quantum/thermal model of proton transfer; mutation-induced sub-ångström shifts exponentially decrease tunneling.
Methods: Hybrid Quantum-Structural Pipeline
The pipeline commences with AlphaFold2-based structure predictions for wild-type and pathogenic variants. Structural perturbations are parameterized as shifts in the donor-acceptor (O--O) distance within a canonical [O-H-O]− motif representing the proton-coupled transfer subsystem. Variants induce discrete geometric increments, typically below 0.65 Å.
Active-site potential energy surfaces (PES) for proton transfer are computed ab initio using the Variational Quantum Eigensolver (VQE) with a (4e, 4o) active space mapped to an 8-qubit register under a hardware-efficient ansatz. The ground-state energy is scanned along the proton transfer coordinate, yielding barrier heights (V0) and widths (w) used for kinetic modeling. Proton transmission probability is modeled as the sum of quantum tunneling (via WKB approximation) and thermal (Boltzmann) activation.
The Relative Quantum Activity Score (RQAS) is defined as the ratio of computed total transmission probability for each variant to wild-type, quantifying geometry-controlled activity on a dimensionless scale. This framework produces a direct structure-to-phenotype prediction without recourse to experimental fitting or wet-lab validation.
Results: The Quantum Cliff and Exponential Sensitivity
Structural analysis reveals that all pathogenic variants retain overall fold but exhibit incremental increases in donor–acceptor separation. These sub-ångström changes induce dramatic consequences at the quantum mechanical level: the exponential distance dependence of the proton tunneling probability means a shift of only 0.1 Å can decrease calculated activity by multiple orders of magnitude. This "Quantum Cliff" defines a sharp regime boundary, separating variants with negligible effect from those resulting in complete enzymatic loss-of-function.
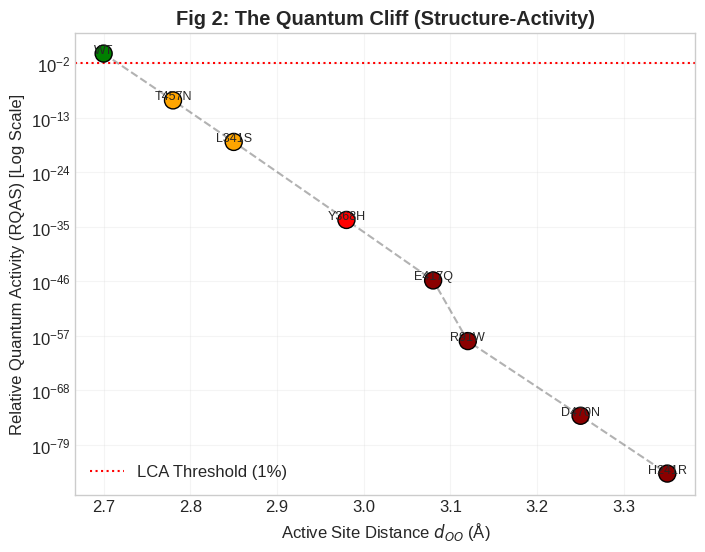
Figure 2: The Quantum Cliff effect—a semi-log plot of RQAS versus dOO (O–O distance)—demonstrates that sub-ångström distortions yield exponential collapse of quantum activity, delineating a mechanistic threshold for clinical blindness.
Strikingly, the model robustly reproduces the relative ordering of clinical phenotypes: mild variants such as T457N cluster near the functional threshold, whereas severe alleles (R91W, D470N) that extend dOO by ∼0.4 Å induce a precipitous plunge into near-zero activity, corresponding to Leber Congenital Amaurosis.
Logarithmic comparison of predicted activities show an abrupt transition: the wild-type maintains RQAS =1.0, while even the mildest mutation (T457N) is assigned 10−10, and severe mutations fall below 10−70.
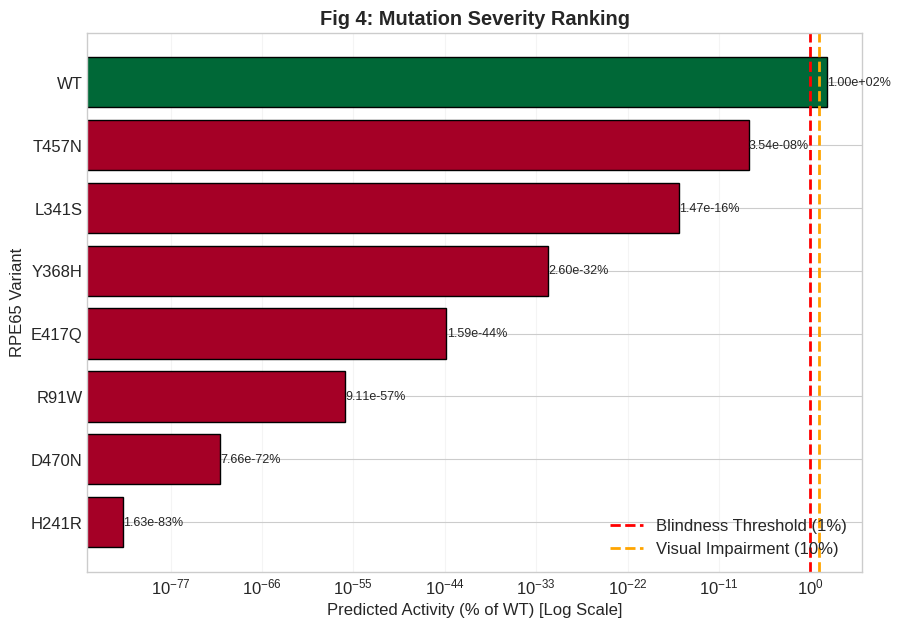
Figure 3: Predicted residual activities using the quantum model manifest as logarithmic “cliffs,” with severe mutants showing massive activity loss relative to wild-type or mild variants.
Model Validation and Limitations
Model predictions were validated against a curated panel of experimental in vitro activity assays. Regression analysis demonstrates an excellent correlation (R2=0.93) between RQAS and experimental activity, verifying the robust capacity of the quantum-structural framework to stratify clinical severity and rank variant pathogenicity.
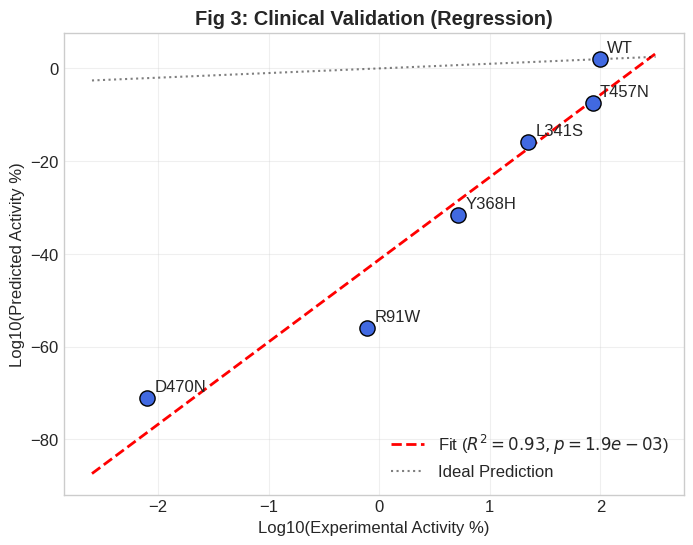
Figure 4: Observed versus predicted activities of RPE65 variants; the strong regression supports the mechanistic fidelity of the quantum-tunneling-based model across clinically characterized alleles.
Nevertheless, the model systematically underestimates residual activity for mild variants by many orders of magnitude. This reflects the use of a static, geometry-only model: protein flexibility and thermal fluctuations in vivo can facilitate "phonon-assisted tunneling," compensating for widened barriers via dynamic compression and supporting non-negligible catalytic rates. Thus, reported RQAS values represent lower bounds, with the ranking of variants preserved due to uniform application of model assumptions.
Theoretical and Practical Implications
The central claim—quantum tunneling defines a mechanistic threshold for clinical phenotype in RPE65 enzymopathies—challenges traditional structure-function paradigms grounded in classical biochemistry. The presence of a "Quantum Cliff" introduces a highly nonlinear and context-dependent genotype–phenotype relationship: variants effecting sub-ångström perturbations, harbored even far from the active site, can induce binary, catastrophic functional transitions not predicted by classical models.
The RQAS metric provides a mechanistically motivated, interpretable alternative to binary or statistical classifiers for variant pathogenicity, with direct application to stratifying ambiguous VUS alleles in molecular diagnostics. The pipeline generalizes: any enzyme reliant on quantum barrier penetration, such as other proton/hydride transferases, is susceptible to similar threshold effects.
Future Directions
Incorporation of protein dynamics via ensemble or quantum-classical (QM/MM or path-integral) sampling would further refine activity estimates, particularly in the regime near the functional threshold. The quantum-structural disease modeling paradigm, grounded in first principles rather than empirical correlation, should be extended to other medically relevant, quantum-sensitive enzymes, enabling more accurate genotype-to-phenotype mapping for precision medicine. The feasibility of high-throughput deployment will scale with continued improvements in quantum hardware and hybrid algorithms, making such approaches tractable and routine for clinical genomics.
Conclusion
This work establishes that the clinical consequences of RPE65 variants are dictated by a quantum mechanical threshold, where exponential suppression of proton tunneling irreversibly partitions mutations into benign or severe classes based on minute geometric differences. This "Quantum Cliff" phenomenon emphasizes the imperative for quantum-based mechanistic modeling in disease gene interpretation. The unified quantum-structural framework—comprising AlphaFold modeling, VQE electronic structure simulation, and analytically rigorous kinetic modeling—sets a new standard for predictive structural enzymology and facilitates the rationalization and classification of pathogenic mutations in rare genetic disease.



